Geanncarlo Lugo-Villarino

CNRS Senior Researcher
I am a senior researcher (Directeur de Recherche, DR2) at the French National Centre for Scientific Research, trained at Harvard University and University of California San Diego. I lead an interdisciplinary program using spatial systems immunology, advanced imaging, and engineered human lung models to uncover how neurovascular and stromal-immune niches reshape macrophage-driven host-pathogen interactions in tuberculosis while building strong France-Argentina collaborations.`
Scientific Background
I am a permanent senior researcher (Directeur de Recherche, DR2) at the French National Centre for Scientific Research (CNRS), leading research at the interface of immunology, host–pathogen interactions, and tissue biology.
I obtained my PhD in Immunology (2000-2006) from Harvard University (Laurie H. Glimcher’s laboratory), where I investigated the transcriptional regulation of dendritic cell (DC) and innate immune responses. I then completed postdoctoral training (2006-2009) at the University of California San Diego (laboratory of David Traver), where I contributed to the identification and characterization of DCs and eosinophils in the zebrafish model. I continued my postdoctoral training (2010-2013) in Olivier Neyrolles’ group at IPBS as an FRM fellow, transitioning toward macrophage-centered mechanisms in Mycobacterium tuberculosis infection. I was recruited to CNRS as CRCN in 2013, obtained my HDR in 2017, and was promoted to Directeur de Recherche (DR2) at CNRS in 2020.
My early independent work contributed to the biology of C-type lectin receptors in the context of tuberculosis (TB) and uncovered how the IL-10/STAT3 and type I interferon pathways contribute to HIV-1 dissemination in TB-skewed macrophages, leading to multiple ANRS-funded projects. As PI of the ANR-funded MMI-TB program, I integrated microbiota, metabolism, and immunity in tuberculosis.
Research Program | Decoding Neurovascular Immune Niches in Tuberculosis
I lead an independent research program focused on understanding how Mycobacterium tuberculosis reshapes the lung environment during infection. Our work goes beyond classical immunology to explore how spatially organized tissue microenvironments influence immune cell behavior and disease progression.
We investigate how resident structural cells of the lung contribute to shaping local immune responses. In particular, we examine how distinct anatomical niches within the lung coordinate macrophage specialization and function during tuberculosis. Our research integrates spatial biology, tissue immunology, and host–pathogen interactions to uncover how the lung landscape is dynamically remodeled in infection.
In particular, we study how stromal-derived resident cells of the lung instruct immune responses. For example, our recent work has identified infection-induced activation of a TUBB3⁺ pericyte-like population within pulmonary granulomas, revealing unexpected neurovascular remodeling programs during TB. By dissecting these tissue-encoded immune circuits, my research aims to redefine host–pathogen interactions at the organ level.
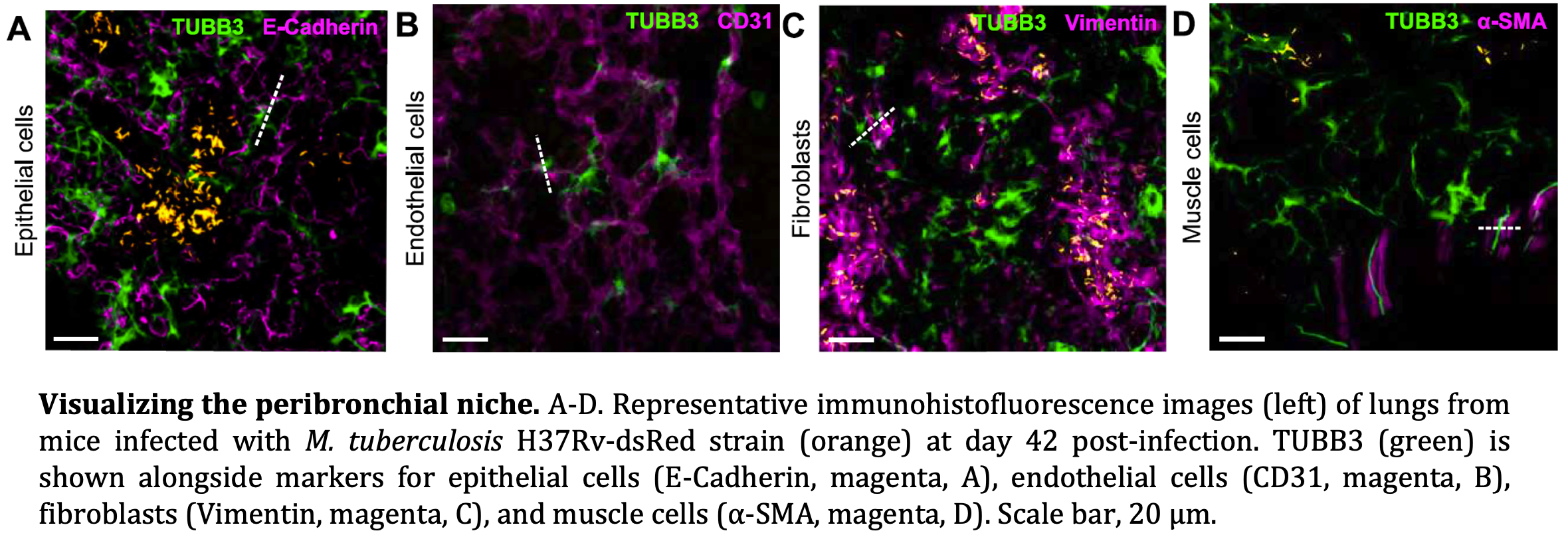
Scientific Approach
My research integrates multi-scale systems immunology with spatial resolution:
• Single-cell RNA sequencing (scRNA-seq)
• Spatial transcriptomics (Visium HD, Xenium)
• Spatial proteomics and mass spectrometry imaging
• 3D lung imaging and tissue clearing
• Advanced genetic mouse models targeting stromal and macrophage subsets
• Human airway organoid systems to model epithelial–immune interactions ex vivo
• Lung organ-on-a-chip platforms to recapitulate dynamic M. tuberculosis infection under controlled mechanical and inflammatory conditions
This integrated pipeline enables us to map and functionally reconstruct how infection dynamically reprograms stromal–immune communication within defined anatomical territories, bridging in vivo spatial resolution with mechanistic validation in engineered human tissue systems.
Leadership and Engagement
I actively coordinate and contribute to international research consortia and collaborative programs across Europe, Latin America, and North America. In particular, I maintain a long-standing strategic collaboration with Dr. Luciana Balboa at INBIRS (University of Buenos Aires, Argentina), formalized through successive bilateral and institutional frameworks including ECOS-SUD (2014–2016), the Laboratoire International Associé (LIA, 2016–2020), and the CNRS International Research Project (IRP, 2021–2025). These programs have strengthened France–Argentina scientific exchange in TB and TB–HIV research, fostering joint publications, student mobility, and coordinated grant applications.
Alongside my research activities, I am deeply committed to mentoring and training the next generation of scientists by supervising Master’s, PhD, and postdoctoral researchers and through capacity-building initiatives linked to international mobility. I regularly contribute to competitive funding proposals, project evaluations, and the publication of scientific articles and reviews, and I engage in international science communication and outreach.

