A new ‘cell saboteur’ against SARS-CoV-2
The inflammation observed in patients infected with SARS-CoV-2, the virus responsible for COVID-19, suggests that pro-inflammatory intracellular complexes, the inflammasomes, regulate the different stages of this infection. Lung epithelial cells express certain inflammasomes and are the main entry point for the virus. The scientists demonstrate that the NLRP1 inflammasome detects SARS-CoV-2 infection in human lung epithelial cells, but behaves as a cell poison that causes cell death and limits the production of infectious virus particles. These results are published in the journal Molecular Cell.
SARS-CoV-2 has infected over 300 million people worldwide. Since it primarily infects cells in the nose, throat and lung, the researchers hypothesised that these cells could potentially control the infection or exacerbate it in individual patients. The inflammation observed in patients infected with SARS-CoV-2 suggests that cellular inflammatory complexes, inflammasomes, are involved in the detection of SARS-CoV-2 in these cells.
In this international study involving French, British, American and Singaporean research teams, the scientists identify the NLRP1 inflammasome as a new detector of SARS-CoV-2, thus participating in its elimination by inducing the death of the infected cell.
To multiply, SARS-CoV-2 expresses the protease NSP5, a viral scissor. In infected cells, NSP5 cuts and activates all the proteins needed to produce new viruses. The researchers show that the cellular protein NLRP1 breaks down this system by mimicking the behaviour of the SARS-CoV-2 proteins. The cutting of NLRP1 by NSP5 leads to its degradation by an intracellular machine, the proteasome. This degradation releases part of NLRP1 which leads to the formation of the NLRP1 inflammasome, a toxic structure for the cell. Thus, the death induced by the NLRP1 inflammasome prevents the replication of SARS-CoV-2 and contributes to protect the organism against this virus.
Taken together, these results identify NLRP1 as a sensor of SARS-CoV-2 infection in lung epithelia and an important antiviral “saboteur” in the body’s fight against this virus. This opens up perspectives in the understanding of lung infections and human susceptibilities to infection.
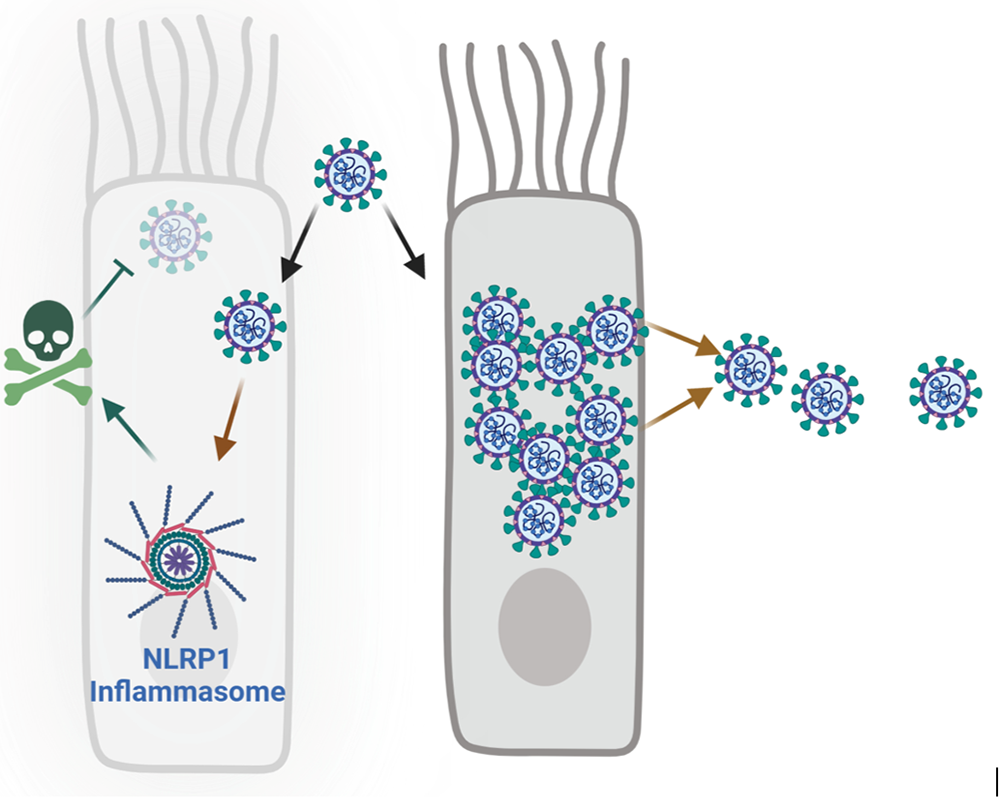
During infection of respiratory cells with SARS-CoV-2, the NLRP1 inflammasome senses the NSP5 protease and induces death of the infected cell, conferring protection against SARS-CoV-2 replication.
© Etienne Meunier, Figure created with Biorender.com (Licence E. Meunier).
Source
“Human NLRP1 is a sensor of pathogenic coronavirus 3CL proteases in lung epithelial cells”
R. Planès, M. Pinilla, K. Santoni, A. Hessel, C. Passemar, K. Lay, P. Paillette, A.-L. Chaves Valadão, K. Samirah Robinson, P. Bastard, N. Lam, R. Fadrique, I. Rossi, D. Pericat, S. Bagayoko, S.-A. Leon-Icaza, Y. Rombouts, E. Perouzel, M. Tiraby, COVID Human Genetic Effort, Q. Zhang, P. Cicuta, E. Jouanguy, O. Neyrolles, C. E Bryant, A. R Floto, C. Goujon, F. Zhong Lei, G. Martin-Blondel, S. Silva, J.-L. Casanova, C. Cougoule, B. Reversade, J. Marcoux, E. Ravet, E. Meunier
Molecular Cell, May 13 2022
Researcher contact
Etienne Meunier | CNRS Research Associate at the Institute of Pharmacology and Structural Biology (IPBS) (CNRS/Université Toulouse III – Paul Sabatier) | Etienne.Meunier@ipbs.fr
Press contact
Françoise Viala |
communication@ipbs.fr | (+33) – 6 01 26 52 59

