Could a well-known antidepressant prevent severe forms of COVID-19?
In the context of the emergence of new variants of SARS-CoV-2, increasingly resistant to vaccines and treatments, the search for complementary therapeutic approaches is important. In vitro and observational studies have recently shown that fluoxetine, known for its antidepressant properties, reduces the clinical signs of COVID-19. Research conducted in part by scientists at the Institute of Pharmacology and Structural Biology and published in the International Journal of Molecular Sciences now confirms an antiviral and anti-inflammatory effect of fluoxetine in the preclinical mouse model of COVID-19.
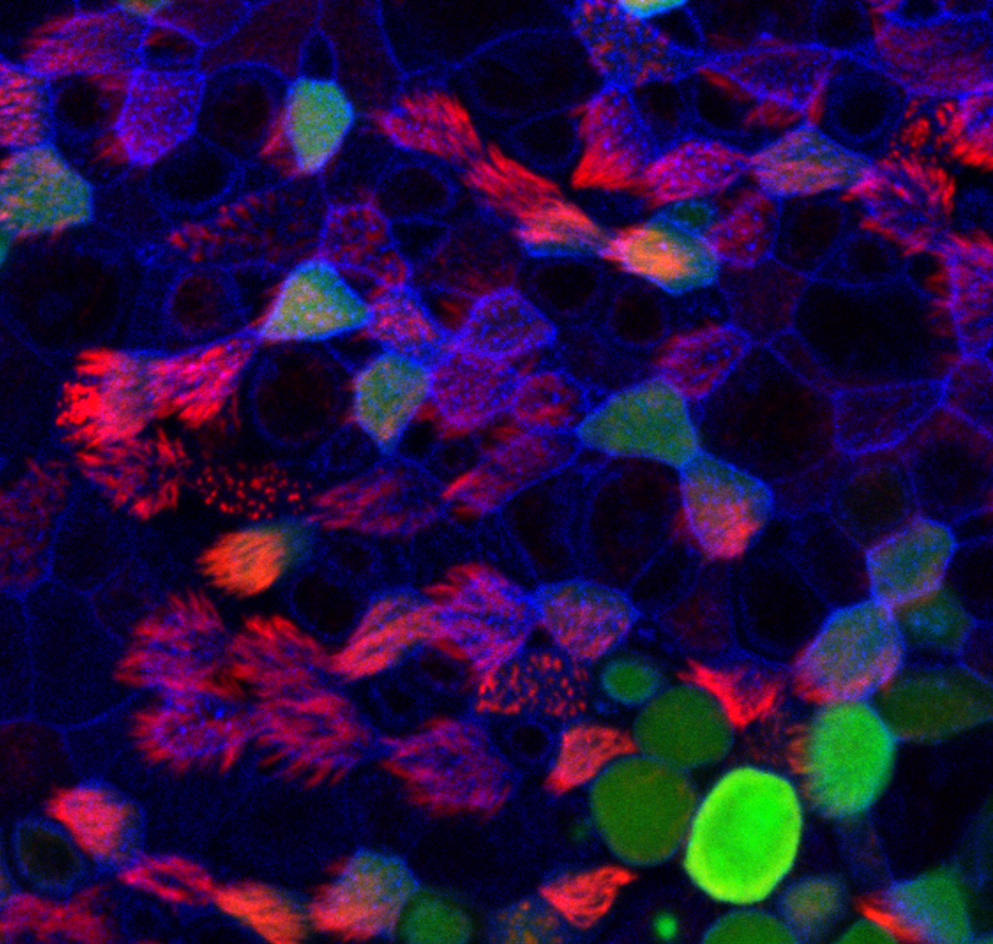
Human air-liquid interface infected with SARS-CoV-2.
Actin filaments appear in blue, cilia in red and virus in green. © Céline Cougoule
This research follows large observational studies that suggest a significant association between fluoxetine use and a lower risk of death in adult patients hospitalized with severe COVID-19. It also reinforces in vitro data showing that modulation of ceramide pathways (a type of lipid contained in the cell wall) would trap the virus’ ACE-2 receptors on the cell surface, thereby inhibiting its entry into target cells and its ability to replicate.
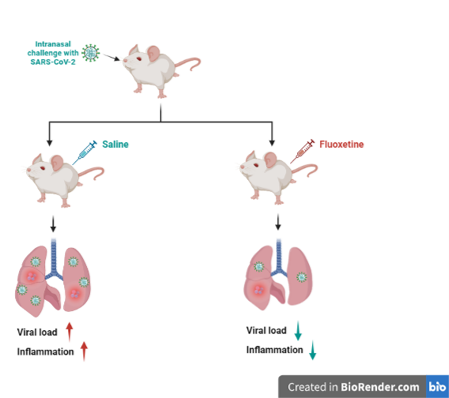
In this article, scientists show that post-infection therapeutic administration of fluoxetine at a dosage modulating the ceramide pathway reduces the viral load in the lungs of treated mice by more than 90% on the second post-infection day, compared to mice treated with saline alone. Furthermore, this active substance attenuates the clinical signs of the disease and strongly reduces the levels of several inflammatory markers. Finally, fluoxetine showed in vitro antiviral activity against different variants of SARS-CoV-2, including the Omicron BA.5 variant.
This preclinical animal model supports in vitro, observational and clinical trial data suggesting that drugs that modulate ceramide pathways – including the two “antidepressant” treatments fluoxetine and fluvoxamine – have an anti-SARS-CoV-2 effect, probably in combination with an anti-inflammatory effect. These results are all the more promising as previous work suggests that these antidepressants can modulate ceramide pathways at a standard dosage in humans. Indeed, fluvoxamine has already been the subject of several randomized controlled clinical trials and cohort studies showing significant clinical benefit in COVID-19 when prescribed at 200-300 mg/d, either as an outpatient or in the ICU. A lack of effect was observed at a lower dosage (100 mg/d) in overweight or obese patients.
© Céline Cougoule
These results need to be confirmed by randomized controlled trials. Large international clinical trials are currently testing the efficacy of fluvoxamine and fluoxetine – including the DNDi-coordinated AntiCoV trial – and are therefore highly anticipated. If positive, they will pave the way for a novel, effective, well-tolerated, and low-cost therapeutic approach to combat COVID-19, particularly in resource-limited countries.
Source
“Antiviral and anti-Inflammatory activities of fluoxetine in a SARS-CoV-2 infection mouse model” David Péricat, Stephen Adonai Leon-Icaza, Marina Sanchez Rico, Christiane Mühle, Iulia Zoicas, Fabian Schumacher, Rémi Planès, Raoul Mazars, Germain Gros, Alexander Carpinteiro, Katrin Anne Becker, Jacques Izopet, Nathalie Strub-Wourgaft, Peter Sjö, Olivier Neyrolles, Burkhard Kleuser, Frédéric Limosin, Erich Gulbins, Johannes Kornhuber, Etienne Meunier, Nicolas Hoertel, Céline Cougoule Int J Mol Sci 2022 Nov 7;23(21):13623. doi: 10.3390/ijms232113623
Researcher’s contact
Céline Cougoule | Celine.Cougoule@ipbs.fr | +33 (0)5 61 17 54 61
Press contact
Françoise Viala | communication@ipbs.fr | +33 (0)6 01 26 52 59

