Deciphering the role of adipose tissue in breast cancer progression and regulation by obesity
Principal investigators
Charlotte Vaysse, Camille Attané and Landry Blanc
Other staff involved
Stéphanie Dauvillier (Engineer), Pascale Bernes-Lasserre (Engineer), Camille Monchatre (PhD Student, Fellow of the Ligue contre le Cancer), Adrien Leeman (PhD Student)
Collaborators
- Dr Anne Bouloumié’s group, Institute of Metabolic and Cardiovascular diseases (I2MC), Toulouse
- Pr Philippe Valet’s group, RESTORE Institute, Toulouse
- Dr Camille Franchet, Pathology Department of the University Toulouse Cancer center (headed by Pr P. Brousset)
- Pr Olivier Piot’group, Translational BioSpectroscopy, Reims
- Dr Justine Ferey, Dr Laurent Debrauwer, Toxalim Platform, INRAE
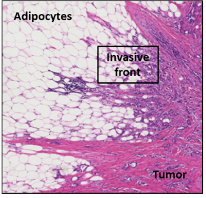
Figure 1 – Adipocytes are major components of the mammary tumoral microenvironment. Hematoxylin/eosin staining of the invasive front in breast cancer. When cancer become invasive, tumor cells (pink/mauve) come into contact with the adipose tissue (white cells).
Breast cancer is the most common cancer in women worldwide. Women with breast cancer who are overweight or obese have a shorter disease-free survival, a higher mortality and a decreased response to chemotherapy, relative to their lean counterparts
Mammary adipose tissue (MAT) is a major component of the breast tumor microenvironment. Our team has been among the first to characterize the modifications of tumor surrounding adipocytes. We named them CAAs for Cancer-Associated adipocytes, a term that is now widely used by the international community (Fig. 1). CAAs exhibit a decrease in size and lipid content and an activated phenotype marked by an increase expression of pro-inflammatory cytokines and proteins involved in the extra-cellular matrix (ECM) remodeling (Dirat et al, Cancer Research, 2011, 500 citations in Web of Science, Top 1%). Upon prolonged exposure to cancer cells, adipocytes will dedifferentiate into fibroblasts and as such represents a sub-population of the Cancer-Associated Fibroblasts (CAFs) (Bochet et al, Cancer Research, 2013, Top 1 % Web of Science). Initially identified by our team in breast cancer, it is now widely acknowledged, in all solid tumors, that the invasion of proximal adipose tissue leads to profound delipidation of adipocytes, that could ultimately lead to the accumulation of fibroblast- like cells. In turns, CAAs promote cancer aggressiveness (especially the invasive capacities of cancer cells) and resistance to conventional chemotherapy through their abilities to secrete different soluble factors including interleukin 6 (Dirat et al, Cancer Research, 2011; Bochet et al, Cancer Research, 2013: Lehuédé et al, Breast Cancer Research, 2019; for review, Duong et al, Oncotarget, 2017).
One of the most specific and emerging mechanism regarding the role of mature adipocytes in the tumor microenvironment involves the ability of cancer cells to advantageously exploit the nourishing role of adipocytes. In breast cancer, we have demonstrated that tumor cells induce lipolysis in adipocytes leading to the release free fatty acid (FFA) that are taken up by tumor cells. These FFA trigger a complex metabolic remodeling in cancer cells (see Figure 2) increasing their invasive and metastatic abilities (Wang et al, JCI insight, Top 1% Web of Science). Again, this metabolic crosstalk has now been demonstrated in a wide range of models such as ovarian, colon and prostate cancers as well as melanoma, and we have recently highlighted the importance of this concept in a review (Attané and Muller, Trends in Cancer, 2020).
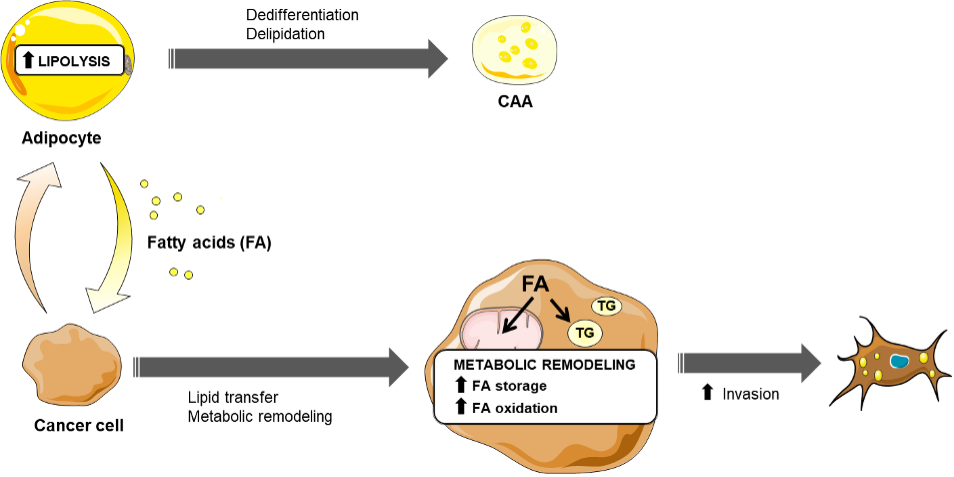
Figure 2. Tumor progression is promoted by tumor cell uptake of lipid released by adipocytes at the invasive front. Schematic representation of the metabolic symbiosis that occurs at the invasive front. Tumor cells release lipolytic signals to transform adipocytes into CAAs. In turns, adipocytes release Fatty Acid (FA). The lipids are internalized and stored as triglycerides (TG) in tumor cells and when needed the tumor cells release FA that are used for fatty acid oxidation (FAO). FAO contributes to increase the invasive abilities of breast cancer cells. Adapted from Attané and Muller. 2020 Trends Cancer. 2020.
Our current hypothesis is that this metabolic symbiosis plays a key role in the tumor-promoting effect of adipocytes. We have recently set up a 3D coculture system of mammary adipocytes obtained from lean and obese patients. This model is used: i) to study the metabolic crosstalk with triple negative breast tumor that appears to exhibit metabolic heterogeneity and ii) to study if this metabolic crosstalk is regulated by obesity.
We also study this metabolic symbiosis directly in tumor microenvironment thank to spatial metabolomic analysis of patient biopsies. The samples are obtained from patients undergoing mastectomy for breast cancers at the “Institut universitaire du cancer Toulouse Oncopole” (IUCT). One of the members of the MICA team at IPBS, Pr Charlotte Vaysse, is a breast cancer surgeon and is in charge of the selection of the patients in collaboration with the Pathology Department of IUCT (Dr Camille Franchet). Then Dr. Landry Blanc implements in the team a multimodal strategy for spatial metabolomic, using mass spectrometry imaging (MSI) guided by immunofluorescence (IF), to decipher spatial distribution of metabolites in complex tissues. The main objective is to study the modification of the lipid content of tumor cells at the tumor invasive front of invasive breast cancers and the nature of these modifications (Figure 3).
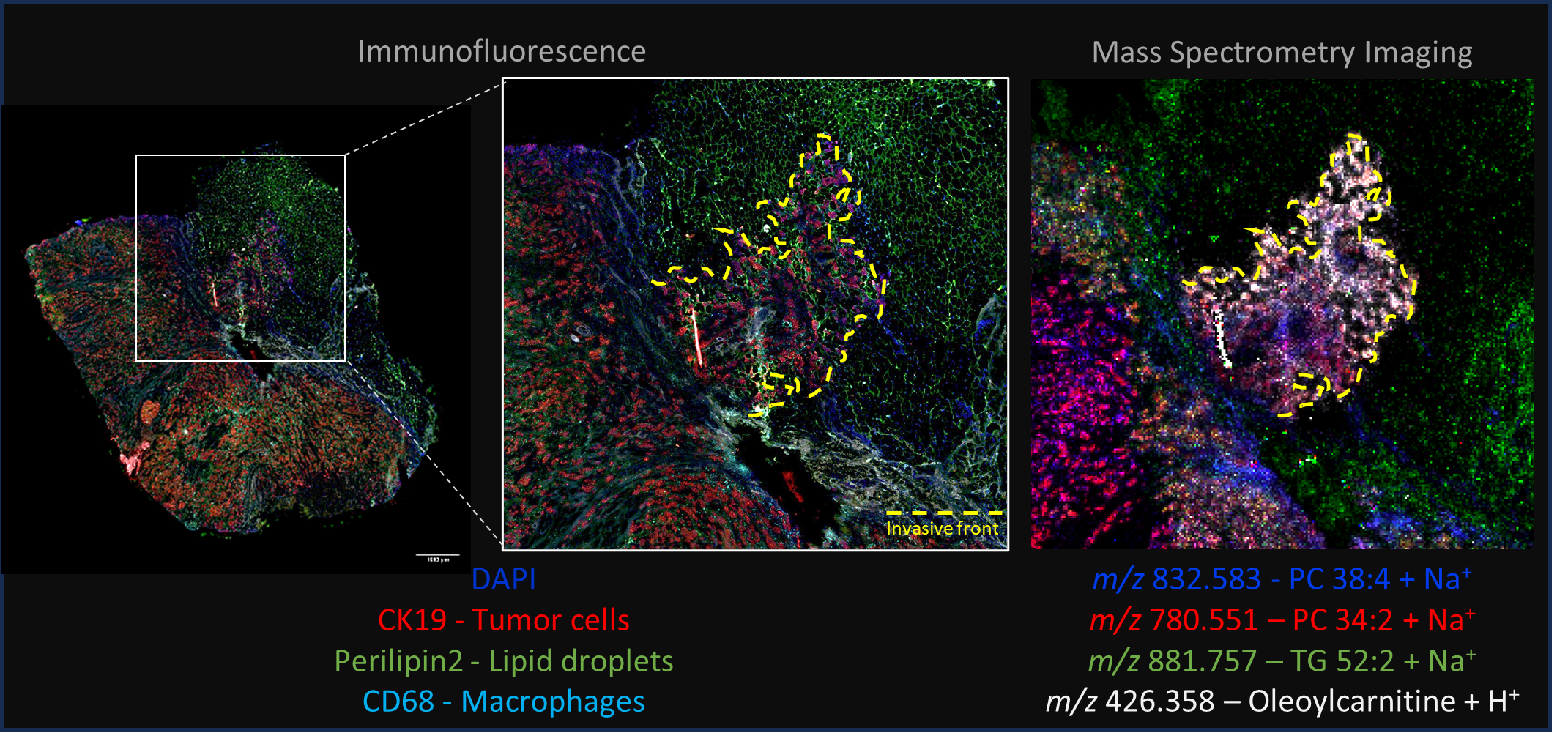
Figure 3 – Multimodal acquisition of Human breast tumor ER+ section to detect spatial distribution of various lipid species. Accumulation of carnitine ester in tumor cells located at invasive front in close contact with adipocyte. MSI and IF staining executed on same tissue section sequentially. MSI was performed in positive mode with CHCA matrix at 20µm lateral resolution in a first time, follow by rinsing and IF staining.
Selected publications
- Dirat B et al. 2011 Cancer-associated adipocytes exhibit an activated phenotype and contribute to breast cancer invasion. Cancer Res (Top 1% Web of Science)
- Bochet L et al. 2011 Cancer-associated adipocytes promote breast tumor radioresistance. Biochem Biophys Res Commun
- Bochet L et al. 2013 Adipocyte-derived fibroblasts promote tumor progression and contribute to the desmoplastic reaction in breast cancer. Cancer Res (Top 1% Web of Science)
- Vaysse C et al. 2017 Inflammation of mammary adipose tissue occurs in overweight and obese patients exhibiting early-stage breast cancer. NPJ Breast Cancer
- Wang YY et al. 2017. Mammary adipocytes stimulate breast cancer invasion through metabolic remodeling of tumor cells. JCI Insight (Top 1% Web of Science)
- Fallone F et al. 2018 Breast cancer, obesity and adipose tissue: a high-risk combination. Med Sci (Paris)
- Lehuédé C et al. 2019 Adipocytes promote breast cancer resistance to chemotherapy, a process amplified by obesity: role of the major vault protein (MVP). Breast Cancer Res

