Dissemination of prostate cancer: a way paved of fat
Principal investigator
Delphine Milhas
Other staff involved
Mathieu Roumiguié (Urologist), Yiyue Jia (PhD student, fellow of the Chinese Research Council), Mathilde Lacombe (MSc student), Stéphanie Dauvillier (Engineer)
Collaborators
- Dr Anne Bouloumié, Institute of Cardiovascular and Metabolic Diseases, Toulouse, France
- Pr Philippe Valet and Dr Cedric Dray, Institute Restore, Toulouse, France
- Pr Philip Scherer, Touchstone Diabetes Center, Dallas, USA
- Dr Sarah Pericart, Pathology Department of the University Toulouse Cancer Center (headed by Pr P. Brousset)
- Surgeons (Dr Mathieu Roumiguié and colleagues), CHU Toulouse
Prostate cancer (PCa) is the second most common form of solid tumor in men worldwide and the fifth leading cause of death from cancer. It is a highly heterogeneous disease, ranging from slow-growing indolent tumors to fatal metastatic carcinomas. Risk stratification is, therefore, the cornerstone for selecting the most appropriate treatment for these men.
The prostate is surrounded by a fat depot named periprostatic adipose tissue (PPAT) that is, like other fat depots, an active endocrine organ. We and others have shown that PPAT contribute to tumor progression through secretion of soluble factors and lipid mediators (reviewed in Estève et al. 2020 Curr Opin Endocr Met Res). Mature adipocytes from PPAT, through an original CCR3/CCL7 axis, favor the homing of cancer cells outside of the prostate gland. This effect is amplified in obesity and controls the local dissemination of prostate cancer in vivo (as shown using animal models and annotated collections of human cancers) (Laurent et al. 2016 Nat Commun). Once tumor cells have invaded the PPAT, FFAs liberated from adipocytes are taken up by tumor cells where they stimulate the expression of pro-oxidant enzymes leading to increased intra-cellular reactive oxygen species and invasive potential of tumor cells. Human tumor analysis revealed that pro-oxidant enzymes expression is highly upregulated at the tumor invasive front where cancer cells are in close proximity to adipocytes, this process being exacerbated by obesity, highlighting the clinical relevance of our results (Laurent et al. 2019 Mol Cancer Res). Our work emphasizes the key role of adipocytes in prostate cancer local dissemination and proposes new molecular targets for the treatment of obese patients exhibiting aggressive diseases. We have reviewed the role of PPAT in prostate cancer dissemination (Estève et al. 2020 Curr Opin Endocr Met Res).
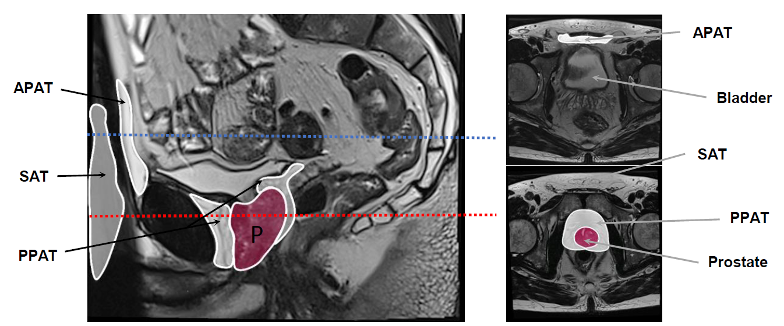
Prostate is surrounded by a specific adipose tissue, the periprostatic adipose tissue (PPAT) as shown on a multiparametric magnetic resonance imaging. Others adipose depots are indicated in addition to PPAT, the sub-cutaneous adipose tissue (SAT) and the abdominopelvic adipose tissue (APAT) which is close to the bladder.
Although changes in PPAT function occur in obesity, many studies have shown that, unlike all other AT, the volume of PPAT, as measured by multiparametric magnetic resonance imaging (mpMRI), does not correlate with BMI. We have recently demonstrated that due to its peculiar vascularization, PPAT exhibits a chronic hypoxic state which induces fibrosis explaining its failure to expand in obesity (Roumiguié et al. 2022 Am J Pathol). Although the volume of PPAT in an individual does not correlate with his BMI, it does vary considerably between individuals. In collaboration with the Urology Department of the Toulouse Cancer Center, we measured on pre-operative mpMRI in a cohort of 351 patients the volume of PPAT. We find tumors specifically from patients with abundant PPAT exhibit several hallmarks of aggressiveness, suggesting that PPAT abundance might be used to improve risk stratification. We also showed that abundant PPAT exhibits extensive extracellular matrix (ECM) remodeling allowing its expansion without inflammation due to the decrease of the mechanical constraints on adipocytes. Moreover, collagen VI degradation in abundant PPAT is associated with production of endotrophin, a matrikin that promotes cancer progression. We find high levels of endotrophin specifically in the urine of patients with abundant PPAT, indicating the clinical relevance of our findings (Esteve et al. 2023 bioRxiv). This part of our work is supported by “La Ligue Nationale Contre le Cancer “ (labeled team).
Selected publications
- Laurent V et al. (2016). Periprostatic adipose tissue acts as a driving force for the local invasion of prostate cancer in obesity: role of the CCR3/CCL7 axis. Nat Commun (Highlights in Nat Rev Cancer Nat Rev Urol, Top 1% Web of Science)
- Laurent V*, Toulet A* et al. (2019). Periprostatic adipose tissue favors prostate cancer cell invasion in an obesity-dependent manner: role of oxidative stress. Mol Cancer Res
- Estève D et al. (2020). Periprostatic adipose tissue a heavy player in prostate cancer progression. Curr Opin Endocr Met Res (invited review)
- Roumiguié M, Estève D et al. (2022). Periprostatic adipose tissue displays a chronic hypoxic state that limits its expandability Am J Pathol
• Esteve D et al. (2023). Collagen remodeling leads to inflammation-free expansion of periprostatic adipose tissue and promotes prostate cancer progression. bioRxiv

