High Endothelial Venules (HEVs), specialized blood vessels for lymphocyte migration
High Endothelial Venules (HEVs) mediate lymphocyte extravasation into lymphoid organs. A decade ago, we discovered that blood vessels with HEV characteristics are also frequently found in solid tumors, in association with cytotoxic T cell infiltration. Contrary to the dogma that blood vessels facilitate tumor growth, tumor-associated HEVs (TA-HEVs) correlated with favorable clinical outcome in breast cancer. Recently, we showed that TA-HEVs function as the main sites of lymphocyte entry in tumors during cancer immunotherapy with anti-PD-1 and anti-CTLA-4 antibodies.
HEVs, portals of entry for lymphocytes into lymphoid organs (Nature 2011, Nat Rev Immunol 2012)
HEVs are anatomically distinct post-capillary venules that function as portals of entry for lymphocytes into lymph nodes and other secondary lymphoid organs (1-3). HEV-mediated recruitment of lymphocytes is a very efficient process since it is estimated that, every second, 5 million lymphocytes migrate through HEVs of the body (1). The endothelial cells lining HEVs have a plump, almost cuboïdal appearance, and express sulfated ligands for lymphocytes, recognized by the HEV-specific antibody MECA-79 (1-3). In 1995, we developed a specific protocol for the isolation of HEV endothelial cells from fresh human lymphoid organs (4) that opened the way to the molecular characterization of these specialized endothelial cells, including the discovery and characterization of 17 novel human genes by our team over the past 20 years (Hevin, PAPSS1, SUT1, SLC26A7, SLC26A11, THAP1 to THAP11, NF-HEV/IL-33).
In 2011, we made a major breakthrough in our understanding of the mechanisms regulating HEV differentiation by demonstrating the critical role of dendritic cells in this process (5). After in vivo depletion of CD11c+ dendritic cells in adult mice, we observed a striking downregulation of HEV-specific marker MECA-79 and HEV-specific genes (GlyCAM-1, FucT-VII, GlcNAc6ST-2) in lymph nodes. Intravital microscopy analyses revealed striking alterations in lymphocyte/HEV interactions, which resulted in an inhibition of HEV-mediated lymphocyte recruitment into lymph nodes (5). Dendritic cells expressed lymphotoxin and dendritic cell-derived lymphotoxin was found to be essential for HEV-mediated lymphocyte recruitment in vivo (5). Together, our findings revealed a previously unsuspected and important function of dendritic cells in the control of HEV phenotype and function.
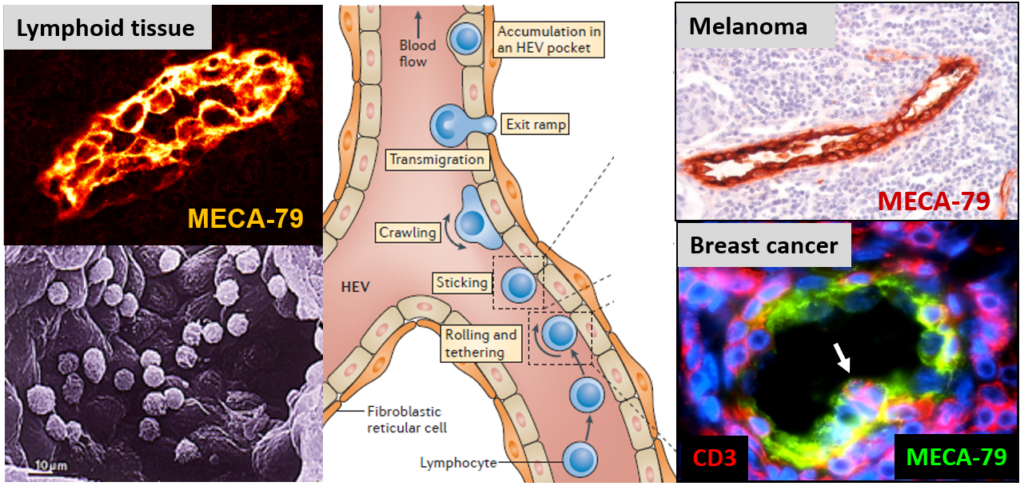
HEVs are specialized blood vessels that mediate lymphocyte migration. HEV endothelial cells in lymphoid organs exhibit a plump morphology, very different from the flat morphology of ‘classical’ endothelial cells. Lymphocytes extravasate through HEVs via a multistep adhesion cascade (rolling, sticking, crawling, transmigration). Tumor-associated HEVs in human melanoma and breast tumors express high levels of the HEV-specific MECA-79 antigens.
Tumor-associated HEVs (TA-HEVs) contribute to the ‘fight’ against cancer (Cancer Res 2011)
We discovered that blood vessels with HEV characteristics (plump endothelial cells expressing HEV-specific marker MECA-79) are frequently found in the stroma of human solid tumors including melanomas, breast, ovarian and lung carcinomas (6-8). In both breast tumors (n = 273) (6, 8) and primary melanomas (n = 225) (7), the density of HEVs was highly correlated with the density of tumor-infiltrating CD3+ and CD8+ cytotoxic T cells, indicating that tumor-associated HEVs (TA-HEVs) may function as major portals of entry for cytotoxic T cells into human solid tumors (Figure 2).
We found that a high density of HEVs in the tumor microenvironment independently conferred a lower risk of relapse and significantly correlated with breast cancer patient longer survival (6). In addition, we found correlations between high densities of TA-HEVs and favourable clinical parameters in primary melanoma (7). Interestingly, TA-HEVs may limit metastasis (probably through their capacity to recruit, into the primary tumor, naïve and memory T cells able to fight tumor cells at distant sites), since a high density of HEVs in breast tumors was associated with longer metastasis-free survival (6). These studies introduced the novel concept that ‘blood vessels in the tumor microenvironment are not all the same and that some types of blood vessels (i.e. TA-HEVs) can contribute to tumor suppression rather than tumor growth’.
This concept could have broad applications for many types of human solid tumors and novel therapeutic strategies based on the modulation of TA-HEVs could have a major impact on clinical outcome of cancer patients.
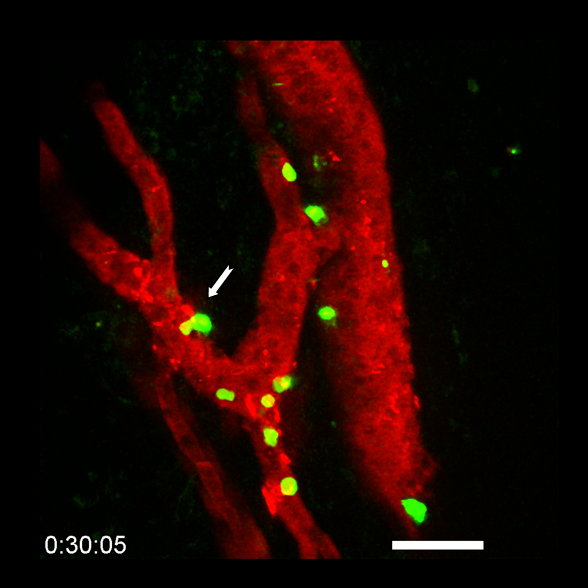
Figure 2: Tumor-associated HEVs (TA-HEVs) function as portals of entry for lymphocytes into tumors during cancer immunotherapy. Multiphoton imaging of lymphocytes (in green) migrating through TA-HEVs (in red) during combination anti-PD-1 plus anti-CTLA-4 immunotherapy. The white arrow indicates a lymphocyte that is leaving the bloodstream and entering the tumor (in black). © E. Bellard and J.-P. Girard.
TA-HEVs mediate lymphocyte entry into tumors and predict response to PD-1 plus CTLA-4 combination immunotherapy (Cancer Cell 2022)
We found that MECA-79+ TA-HEVs function as major sites of lymphocyte extravasation into tumors during cancer immunity and immunotherapy with dual immune checkpoint blockade (ICB; anti-CTLA-4 plus anti-PD-1 antibodies). Intravital imaging revealed that lymphocytes arrested in the tumor microcirculation almost exclusively in TA-HEVs, both at baseline and during ICB (9). We found that TA-HEVs correspond to the most inflamed post-capillary venules of tumors, and are associated with increased infiltration of various T cell subsets, including stem-like CD8+ T cells. Furthermore, we showed that increasing the frequency of TA-HEVs, improved the efficacy of combined ICB. Together, our data suggested that TA-HEVs are the main sites of entry into tumors for various subsets of lymphocytes critical for anti-tumor immunity and response to ICB. In collaboration with Caroline Robert (Gustave Roussy), we next quantified MECA-79+ TA-HEVs in pre-treatment metastatic melanoma lesions in a large cohort (n=93) of patients undergoing ICB therapy, and found that high numbers of TA-HEVs predicted both treatment response and patient survival, particularly for patients treated with combined ICB (anti-PD-1 plus anti-CTLA-4 antibodies). In summary, this study demonstrated the critical role of TA-HEVs in lymphocyte entry into tumor at baseline and during ICB therapy, and indicated that TA-HEVs represent attractive targets for cancer diagnosis and treatment.

