Immune Detection and Elimination of Pathogens

Group Leader
Over the last 20 years, cell deaths emerged as crucial processes driving inflammation, host defense against infections and pathologies. To understand the determinants of various cell deaths, microbial interactions with cells are a fantastic field of investigation as pathogens use virulence factors to modify the regulation of various host-derived cell death pathways.
The group aims to expand and strengthen the knowledge already developed but also to tackle the challenges of tomorrow in the field of cell death, immunity, host pathogen interactions but also environmental exposure. Indeed, during infections, strongly influencing the vital prognostic of patients are the intrinsic microbial characteristics as well as environmental, age, sex and genetic parameters.

Inflammasomes are intracellular multi-protein complexes that play essential functions in immunity against pathogens. Specifically, inflammasomes both promote a regulated form of cell death termed pyroptosis and trigger the release of the key inflammatory cytokines interleukin (IL)-1β and IL-18, two important processes in controlling microbial infections. Because of their involvement in various pathophysiological conditions, our understanding of inflammasomes has generated an ever-growing interest in the scientific and medical communities over the last 20 years. Yet, most of this knowledge comes either from the use of rodent models, which may lack the specific structural and functional characteristics of human inflammasomes, or from the use of immune cells, which express a wide range of inflammasomes but lack inflammasomes specifically enriched in non-immune cells.
Thus, we study crucial and yet unexplored functions of unique human inflammasomes in epithelia (skin & corneal keratinocytes, airways, lung & intestinal epithelial cells) and in other compartments (e.gs neurons, endothelial cells). We further address inflammasome regulation and functions in atypical and yet understudied immune cells, namely the granulocytes neutrophils and eosinophils as well as in mast cells, critical sentinels of the immune responses.
To address these issues, we use a combination of technologies in microbiology, immunology, biochemistry, molecular and cell biology in various human cellular models developed in the group, including primary human epithelial organoids and organ on a chip.
This multidisciplinary research generates major discoveries in the regulation of human inflammasomes and their interactions with microbial pathogens and various environmental threats.
Contacts for interested students, post-docs, technicians/engineers, researchers, lecturers and others :
etienne.meunier@ipbs.fr
celine.cougoule@ipbs.fr
Team members
Research Scientists
Céline Cougoule (CNRS)
Raoul Mazars (University of Toulouse)
Etienne Meunier (CNRS)
Research Engineers
David Pericat (CNRS)
Chloé Rives
Postdoctoral Fellow
Thomas Benoist
Caio Bomfim
PhD Students
Léa Fromont
Andréa Gomes
Margaux Paradis
Léa Ravon-Katossky
Romain Vergé
Lylia Hakem
Embedded Emerging Research Team
Virginie Stévenin (Emerging PI – FRM)
Our research projects
To unlock the immune response: The cellular and molecular means by which intracellular sensors detect pathogens
To shortcut the intracellular PRRs: Non-canonical functions of cytosolic-derived sensors
Stromal immunity: To decipher the immune and microbicidal responses of the epithelia
Gorse et al. (2025) Portimine A toxin causes skin inflammation through ZAKα-dependent NLRP1 inflammasome activation.Cell Rep
Blot et al. (2024) Leishmania infantum exploits the anti-ferroptosis effects of Nrf2 to escape cell death in macrophages. Mol Cell
Rozario et al. (2024) Mechanistic basis for potassium efflux-driven activation of the human NLRP1 inflammasome. Proc Natl Acad Sci USA
Pinilla et al. (2023) EEF2-inactivating toxins engage the NLRP1 inflammasome and promote epithelial barrier disruption. J Exp Med
Leo-Icaza et al. (2023) Druggable redox pathways against Mycobacterium abscessus in cystic fibrosis patient-derived airway organoids. PLoS Pathog
Planès et al. (2022) Human NLRP1 is a sensor of pathogenic coronavirus 3CL proteases in lung epithelial cells. Mol Cell
Iakobachvili et al. (2022) Mycobacteria-host interactions in human bronchiolar airway organoids. Mol Microbiol.
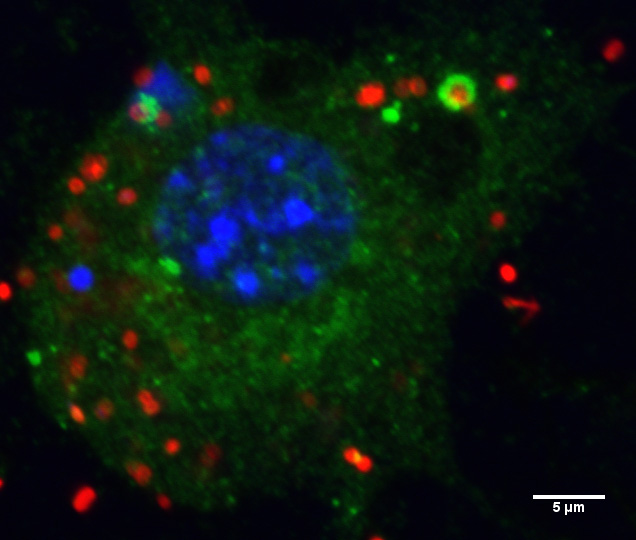
Murine macrophage infected with Francisella novicida (Red) and stained for LC3 (Green).
Collaborations
Olivier Neyrolles, IPBS institute, Toulouse, France
Julien Buyck, Univ. of Poitiers, France
Jessica Quintin, Pasteur Institute, Paris, France
Patrizia D’Adamo, San Rafaele institute, Milan, Italy
Petr Broz, Univ. of Lausanne, Lausanne, Switzerland
Peter J. Peters, Maastricht MultiModal Molecular Imaging Institute (M4I), Maastricht, Netherlands
Hans Clevers, Hubrecht Institute, Utrecht, Netherlands
Funding
The team is supported by several grants and funding from:
– The European Research Council (ERC Starting Grant 2019-2023)
– Agence Nationale pour la Recherche (2019-2022)
– ATIP-Avenir (2017-2020), young team leader program
– Fondation pour la Recherche Médicale (2016-2019), «Amorçage de jeunes équipes » program
– University of Toulouse
– Fondation Fonroga
– Partnership Hubert Curien (PHC) VAN GOGH 2018

2025
Gorse L, Plessis L, Wearne S, Paradis M, Pinilla M, Chua R, Lim SS, Pelluz E, Toh GA, Mazars R, Bomfim C, Hervé F, Lhaute K, Réveillon D, Suire B, Ravon-Katossky L, Benoist T, Fromont L, Péricat D, Neil Mertens K, Derrien A, Terre-Terrillon A, Chomérat N, Bilien G, Séchet V, Carpentier L, Fall M, Sonko A, Hakim H, Sadio N, Bourdeaux J, Cougoule C, Henras AK, Perez-Oliva AB, Brehmer P, Roca FJ, Zhong FL, Common J, Meunier E*, Hess P.* * shared study lead. Portimine A toxin causes skin inflammation through ZAKα-dependent NLRP1 inflammasome activation. EMBO Mol Med. 2025 Mar;17(3):535-562. doi: 10.1038/s44321-025-00197-4. Epub 2025 Feb 13.
2024
Blot C, Lavernhe M, Lugo-Villarino G, Coulson K, Salon M, Tertrais M, Planès R, Santoni K, Authier H, Jacquemin G, Rahabi M, Parny M, Letron IR, Meunier E*, Lefèvre L*, Coste A*. * shared study lead Leishmania infantum exploits the anti-ferroptosis effects of Nrf2 to escape cell death in macrophages. Cell Rep. 2024 Sep 24;43(9):114720. doi: 10.1016/j.celrep.2024.114720. Epub 2024 Sep 7.
Rozario P, Pinilla M, Gorse L, Vind AC, Robinson KS, Toh GA, Firdaus MJ, Martínez JF, Kerk SK, Lin Z, Chambers JC, Bekker-Jensen S, Meunier E*, Zhong F*. * shared study lead. Mechanistic basis for potassium efflux-driven activation of the human NLRP1 inflammasome. Proc Natl Acad Sci U S A. 2024 Jan 9;121(2):e2309579121. doi: 10.1073/pnas.2309579121. Epub 2024 Jan 4.
2023
Pinilla M, Mazars R, Vergé R, Gorse L, Paradis M, Suire B, Santoni K, Robinson KS, Toh GA, Prouvensier L, Leon-Icaza SA, Hessel A, Péricat D, Murris M, Guet-Revillet H, Henras A, Buyck J, Ravet E, Zhong FL, Cougoule C, Planès R, Meunier E. EEF2-inactivating toxins engage the NLRP1 inflammasome and promote epithelial barrier disruption. J Exp Med. 2023 Oct 2;220(10):e20230104. doi: 10.1084/jem.20230104. Epub 2023 Aug 29.
Leon-Icaza SA, Bagayoko S, Vergé R, Iakobachvili N, Ferrand C, Aydogan T, Bernard C, Sanchez Dafun A, Murris-Espin M, Mazières J, Bordignon PJ, Mazères S, Bernes-Lasserre P, Ramé V, Lagarde JM, Marcoux J, Bousquet MP, Chalut C, Guilhot C, Clevers H, Peters PJ, Molle V, Lugo-Villarino G, Cam K, Berry L, Meunier E, Cougoule C. Druggable redox pathways against Mycobacterium abscessus in cystic fibrosis patient-derived airway organoids. PLoS Pathog. 2023 Aug 24;19(8):e1011559. doi: 10.1371/journal.ppat.1011559. eCollection 2023 Aug.
2022
Planès R, Pinilla M, Santoni K, Hessel A, Passemar C, Lay K, Paillette P, Valadão AC, Robinson KS, Bastard P, Lam N, Fadrique R, Rossi I, Pericat D, Bagayoko S, Leon-Icaza SA, Rombouts Y, Perouzel E, Tiraby M; COVID Human Genetic Effort; Zhang Q, Cicuta P, Jouanguy E, Neyrolles O, Bryant CE, Floto AR, Goujon C, Lei FZ, Martin-Blondel G, Silva S, Casanova JL, Cougoule C, Reversade B, Marcoux J, Ravet E, Meunier E. Human NLRP1 is a sensor of pathogenic coronavirus 3CL proteases in lung epithelial cells. Mol Cell. 2022 Jul 7;82(13):2385-2400.e9. doi: 10.1016/j.molcel.2022.04.033. Epub 2022 May 16.
Iakobachvili N, Leon-Icaza SA, Knoops K, Sachs N, Mazères S, Simeone R, Peixoto A, Bernard C, Murris-Espin M, Mazières J, Cam K, Chalut C, Guilhot C, López-Iglesias C, Ravelli RBG, Neyrolles O, Meunier E, Lugo-Villarino G, Clevers H, Cougoule C*, Peters PJ*. * shared study lead. Mycobacteria-host interactions in human bronchiolar airway organoids. Mol Microbiol. 2022 Mar;117(3):682-692. doi: 10.1111/mmi.14824. Epub 2021 Nov 5.
Santoni K, Pericat D, Gorse L, Buyck J, Pinilla M, Prouvensier L, Bagayoko S, Hessel A, Leon-Icaza SA, Bellard E, Mazères S, Doz-Deblauwe E, Winter N, Paget C, Girard JP, Pham CTN, Cougoule C, Poincloux R, Lamkanfi M, Lefrançais E, Meunier E, Planès R. Caspase-1-driven neutrophil pyroptosis and its role in host susceptibility to Pseudomonas aeruginosa. PLOS Pathog 2022 Jul 18;18(7):e1010305. doi: 10.1371/journal.ppat.1010305
Planès R, Pinilla M, Santoni K, Hessel A, Passemar C, Lay K, Paillette P, Valadão AC, Robinson KS, Bastard P, Lam N, Fadrique R, Rossi I, Pericat D, Bagayoko S, Leon-Icaza SA, Rombouts Y, Perouzel E, Tiraby M; COVID Human Genetic Effort; Zhang Q, Cicuta P, Jouanguy E, Neyrolles O, Bryant CE, Floto AR, Goujon C, Lei FZ, Martin-Blondel G, Silva S, Casanova JL, Cougoule C, Reversade B, Marcoux J, Ravet E, Meunier E. Human NLRP1 is a sensor of pathogenic coronavirus 3CL proteases in lung epithelial cells. Mol Cell 2022 Jul 7;82(13):2385-2400.e9. doi: 10.1016/j.molcel.2022.04.03
2021
Bagayoko S, Leon-Icaza SA, Pinilla M, Hessel A, Santoni K, Péricat D, Bordignon PJ, Moreau F, Eren E, Boyancé A, Naser E, Lefèvre L, Berrone C, Iakobachvili N, Metais A, Rombouts Y, Lugo-Villarino G, Coste A, Attrée I, Frank DW, Clevers H, Peters PJ, Cougoule C, Planès R, Meunier E. Host phospholipid peroxidation fuels ExoU-dependent cell necrosis and supports Pseudomonas aeruginosa-driven pathology. PLOS Pathog 2021 Sep 13;17(9):e1009927. doi: 10.1371/journal.ppat.1009927
2020
Eren E, Planès R, Bagayoko S, Bordignon PJ, Chaoui K, Hessel A, Santoni K, Pinilla M, Lagrange B, Burlet-Schiltz O, Howard JC, Henry T, Yamamoto M, Meunier E.Irgm2 and Gate-16 cooperatively dampen Gram-negative bacteria-induced caspase-11 response. EMBO Rep 2020 Nov 5;21(11):e50829. doi: 10.15252/embr.202050829
2018
Santos JC, Dick MS, Lagrange B, Degrandi D, Pfeffer K, Yamamoto M, Meunier E, Pelczar P, Henry T, Broz P. LPS targets host guanylate-binding proteins to the bacterial outer membrane for non-canonical inflammasome activation. EMBO J 2018 Mar 15;37(6):e98089. doi: 10.15252/embj.201798089
2015
Meunier E, Wallet P, Dreier RF, Costanzo S, Anton L, Rühl S, Dussurgey S, Dick MS, Kistner A, Rigard M, Degrandi D, Pfeffer K, Yamamoto M, Henry T, Broz P. Guanylate-binding proteins promote activation of the AIM2 inflammasome during infection with Francisella novicida. Nat Immunol 2015 May;16(5):476-484. doi: 10.1038/ni.3119
Salimata Bagayoko | Clinical Research scientist, Aubervilliers, France
Pierre-Jean Bordignon | Engineer CNRS, Toulouse, France
Elif Eren | Assistant professor, Univ. Istanbul, Turkey
Léana Gorse | Post-doctoral fellow, Université de Lausanne, Suisse
Audrey Hessel | Tenured Engineer Institut Pasteur, Paris, France
Stephen Leon Icaza | Post-doctoral fellow, University of Cambridge, UK
Remi Planès | Project Manager, Invivogen, Toulouse, France
Miriam Pinilla | Post-doctoral fellow, Insituto Murciano de Investigacion Biosanitaria, Spain
Karin Santoni | Post-doctoral fellow, Imperial College, London, UK
Ronald Siaden-Ortega | Post-doctoral fellow, Strasbourg, France
Bastien Suire | Gap year

