Interleukin-33 (IL-33), an alarm signal with crucial roles in innate immunity, inflammation and allergy
Interleukin-33 (IL-33) (1-4) is a nuclear cytokine from the IL-1 family abundantly expressed in blood vessels and barrier tissues (lungs, stomach, skin) that was initially discovered by our team as a nuclear factor of high endothelial venules (NF-HEV) in lymphoid organs (5, 6). IL-33 functions as an alarm signal (alarmin) released upon tissue damage to alert various types of immune cells. It plays critical roles in tissue homeostasis and repair, type 2 immunity, viral infection, and allergic and non-allergic inflammation (1-3).
IL-33, a cytokine from the IL-1 family
Similarly to IL-1, IL-33 (270 aa, 30.7 kDa) is synthesized as a 31 kDa protein that lacks a clear signal peptide (2). It exhibits at its carboxy-terminus a single structural domain formed from 12 beta strands, arranged in a so-called IL-1/FGF beta-trefoil fold (2). IL-33 activates Myd88-dependent signalling pathways in immune cells expressing the ST2/IL-1RAcP receptor complex, including many cell types involved in type-2 immunity such as group 2 innate lymphoid cells (ILC2s, natural helper cells, nuocytes, innate helper 2 cells; a novel lineage negative innate lymphoid cell type), mast cells and their progenitors, basophils, eosinophils and Th2 cells (1-3). Other targets of IL-33 include macrophages, dendritic cells, certain subsets of regulatory T cells, NK and iNKT cells, neutrophils, Th1 cells and CD8+ T cells (2, 3). ILC2s, which produce extremely high amounts of the Th2 cytokines IL-5 and IL-13 in response to IL-33 and play critical roles in type-2 immunity and eosinophil homeostasis, are major targets of IL-33 in vivo (1, 2).
IL-33, a tissue-derived alarm signal regulated by proteases
We showed that endogenous IL-33 is constitutively expressed to high levels in the nuclei of producing cells during homeostasis, including epithelial cells from various barrier tissues, endothelial cells from blood vessels, and fibroblastic reticular cells of lymphoid organs (6-8). Nuclear localization of IL-33 is a fundamental property of the protein that has been observed in all producing cells, both in human and mouse tissues (6-8). In the nucleus, IL-33 associates with chromatin through a short chromatin-binding motif in its N-terminal nuclear domain, that recognizes the heterodimer formed by histones H2A and H2B (9). Although already high during homeostasis, expression of IL-33 is further upregulated during inflammation, and the protein can be produced by additional cell types (1-4, 8). We have shown that full-length IL-33 is biologically active and that it can be released from the nucleus of producing cells after cellular damage or necrotic cell death (10). We thus proposed that IL-33 may function as an alarm signal (alarmin) to alert cells of the innate immune system of tissue injury during trauma or infection (1, 3, 4, 7, 10).
We demonstrated that IL-33 cytokine activity is regulated by proteolytic maturation (1, 3, 4). Apoptotic caspases inactivate IL-33 whereas inflammatory proteases process IL-33 into shorter forms with increased biological activity (1, 10-12). The two major forms generated by neutrophil and mast cell proteases, IL-33(95-270) and IL-33(109-270), have a ~ 30-fold higher activity than full length IL-33(1-270) for activation of ILC2s ex vivo, and are highly potent inducers of type-2 responses in vivo (11, 12).
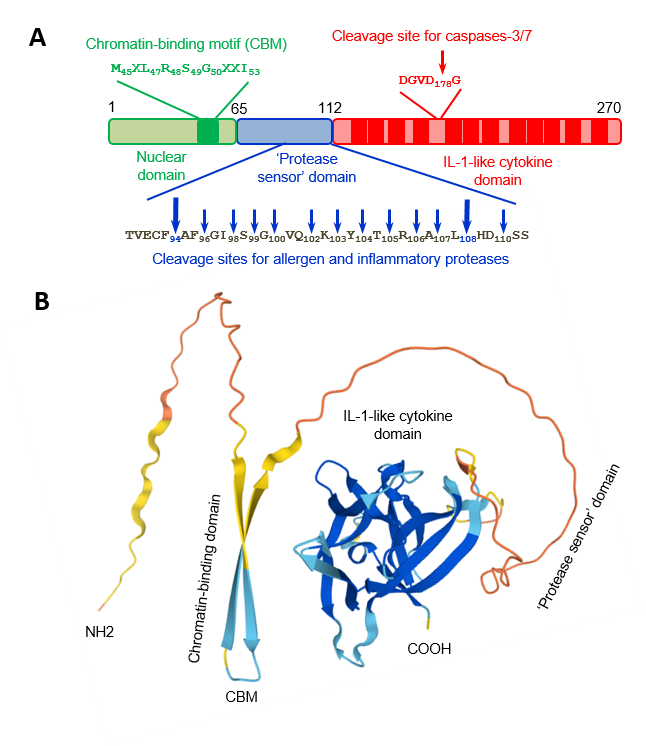
The IL-33 protein. A) Human IL-33 protein is composed of three domains. Chromatin-binding motif and cleavage sites for caspases, allergen proteases, and inflammatory proteases are indicated. B) Three dimensionnal structure of the mouse IL-33 protein predicted by Alphafold.
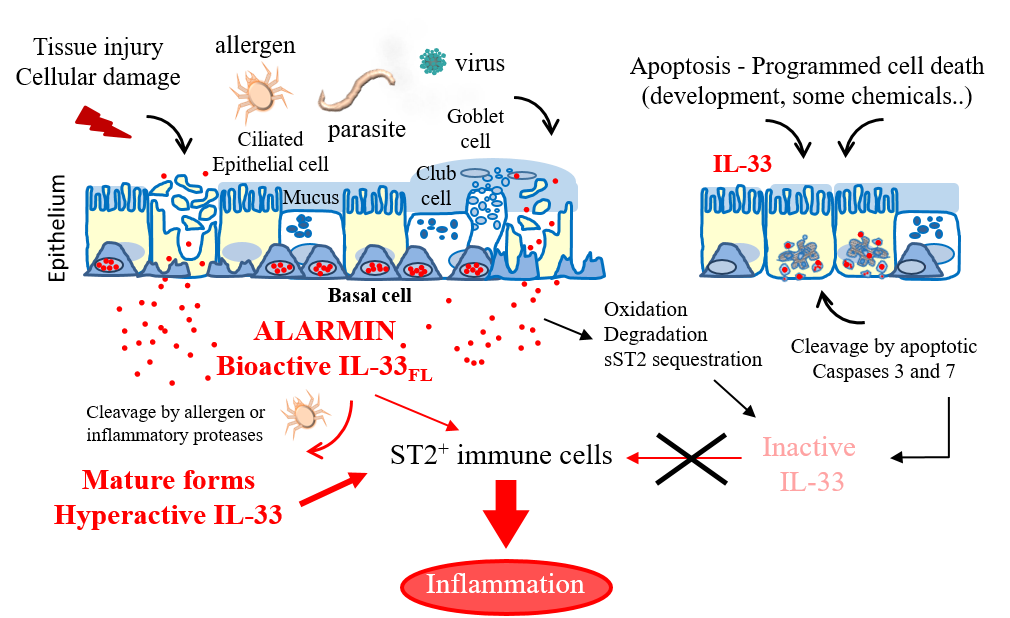
IL-33 alarmin in lung inflammation. IL-33 is released extracellularly upon tissue damage, following exposure to allergens or infection. After release, IL-33 ‘raises the alarm’ in the immune system by activating various types of immune cells expressing the IL-33 receptor ST2. Mechanisms regulating IL-33 bioactivity are indicated.
The IL-33/ILC2 axis in allergic inflammation and human asthma
IL-33 induces allergic airway inflammation by stimulating lung ILC2s. Indeed, IL-33/ST2 signaling has been shown to be required for IL-5 and IL-13 production by lung ILC2s and airway eosinophilia, following exposure to allergens (1, 2). IL-33 and ILC2s are also important for allergic inflammation in other tissues (skin, nasopharynx). IL-33-responsive ILC2s have been shown to be enriched in lesional skin biopsies of atopic dermatitis patients and in nasal polyps of patients with chronic rhinosinusitis (1, 2).
Interestingly, genetic studies suggest a major role for the IL-33/ST2 pathway in human asthma. Indeed, IL-33 and ST2 were the only two genes recurrently identified in all major genome-wide association studies of asthma, which included thousands of patients from diverse ethnic groups and different forms of asthma (asthma associated with blood eosinophils, early childhood asthma with severe exacerbations, …) (1). The high levels of constitutive expression of IL-33 in epithelial barriers of the lungs and its ability to be released very rapidly (minutes) after allergen challenge for ‘alarming’ ILC2s and other immune cells are likely to contribute to the importance of the IL-33/ILC2 axis in asthma.
Recently, we reported an important breakthrough in the understanding of the molecular mechanisms involved in the initiation of allergic inflammation. We discovered a critical role of IL-33 as a protease sensor that is cleaved and activated by proteolytic activities associated with clinically relevant aeroallergens and allergens involved in occupational asthma (13). Interfering with this IL-33-based protease sensing mechanism could represent a new approach for reducing allergic responses in asthma and other allergic diseases.

