IP6: an endogenous small molecule which promotes DNA repair
IP6, inositol hexaphosphate or phytic acid, is a product of cellular metabolism that binds to numerous proteins whose functions it regulates. In an article published in the journal Nucleic Acids Research, scientists reveal how IP6 stabilizes the assembly of a complex responsible for DNA break repair in humans.
Many cancer therapies induce DNA double-strand breaks. A DNA double-strand break represents the most serious type of DNA damage to a cell, since it amounts to splitting a chromosome in two. The resulting chromosome fragments can be lost or lead to translocations if they are not quickly rejoined. Unrepaired breaks most often lead to cell death, a property exploited to eradicate tumor cells during radiotherapy or certain chemotherapies.
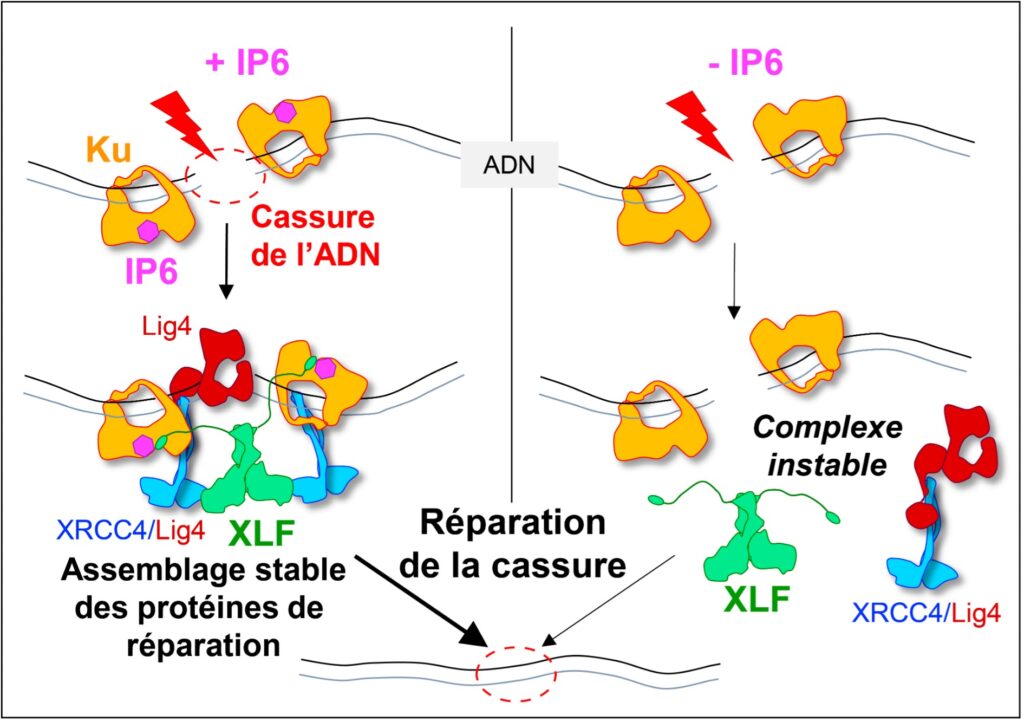
Yet DNA double-strand break repair pathways exist, and their performance in tumors determines the efficacy of these therapies. The dominant system for repairing DNA double-strand breaks in human cells is Non-Homologous End Joining, or NHEJ, initiated by the Ku protein, which rapidly encircles the ends of the break and acts as a hub for the other proteins needed to weld these ends together. Using two techniques for studying proteins at atomic scale (crystallography and cryo-electron microscopy), the scientists discovered how a small molecule produced by the body and sometimes used as a dietary supplement, inositol-hexaphosphate (IP6) or phytic acid, binds to the Ku protein.
The Ku protein, an interesting target for killing tumor cells
The scientists then analyzed the effect of IP6 on break repair in human cells. They modified the Ku protein to block its binding to IP6, and thus understand how the latter acts. The Ku mutants revealed that the presence of IP6 on the Ku protein stimulates its binding to the XLF protein, an essential step in efficiently welding the DNA break (see the model below). IP6 thus plays a key role in stabilizing the large complex of NHEJ proteins required for DNA break repair. This fundamental work resolves the question raised two decades ago regarding the role of IP6 in DNA double-strand break repair. It also opens up therapeutic prospects by identifying a new region on the Ku protein that could be targeted by small molecules to block DNA break repair in tumor cells.
Reference
Structural and functional basis of inositol hexaphosphate stimulation of NHEJ through stabilization of Ku-XLF interaction
Antonia Kefala Stavridi§, Amandine Gontier§, Vincent Morin§, Philippe Frit §,Virginie Ropars, Nadia Barboule, Carine Racca, Sagun Jonchhe, Michael J. Morten, Jessica Andreani, Alexey Rak, Pierre Legrand, Alexa Bourand-Plantefol, Steven W. Hardwick, Dimitri Y. Chirgadze, Paul Davey, Taiana Maia De Oliveira, Eli Rothenberg, Sébastien Britton*, Patrick Calsou*, Tom L. Blundell*, Paloma F. Varela*, Amanda K. Chaplin* and J.B Charbonnier* Nucleic Acids Res 2023 doi : 10.1093/nar/gkad863
Contact (IPBS)
Patrick Calsou (Patrick.Calsou@ipbs.fr)
Sébastien Britton (Sebastien.Britton@ipbs.fr)
Press
Francoise Viala (Communication@ipbs.fr)