Metal efflux platforms in the membrane of bacteria
Scientists have discovered a novel mechanism of resistance to metal intoxication in macrophages involving mobile platforms in the plasma membrane of the bacterium in the pathogen Mycobacterium tuberculosis, the agent of tuberculosis. These platforms consist of metal efflux pumps of the “P-type ATPase” family, as well as an entirely new class of proteins, the “PacL” proteins. Homologues of PacL proteins are present in many bacteria, and the presence of toxic metal efflux platforms is probably widespread in prokaryotes. These results, which shed light on the metallobiology of bacteria, are published in the journal Nature Communications.
Bacteria, whether environmental, commensal, symbiotic, opportunistic or pathogenic, must adapt to their constantly changing environment. When pathogenic bacteria infect their hosts, they are phagocytosed by macrophages, sentinel immune cells that patrol all tissues and ensure their cleaning and thus the balance.
Scientists had previously identified a mechanism by which macrophages can eliminate or control the pathogens they ingest through phagocytosis, by accumulating metals, particularly zinc, in the phagocytosis vacuoles at concentrations potentially toxic to bacteria. They had also demonstrated the role of the metal efflux pump CtpC, a pump of the P-type ATPase family, in the resistance of the tuberculosis bacillus, Mycobacterium tuberculosis, to metal stress in macrophages. The CtpC protein is produced together with a small protein with a domain of unknown function (DUF1489), called Rv3269. Like CtpC, the Rv3269 protein locates in the plasma membrane of the bacteria. It also carries a metal-binding motif.
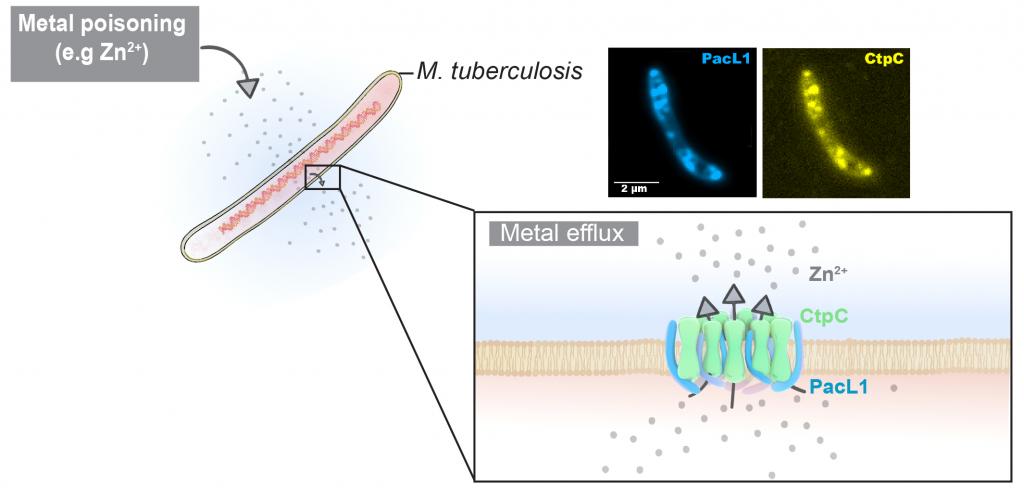
In the present study, the scientists show that CtpC and Rv3269 are located in mobile microdomains in the bacterial membrane, where Rv3269 can both bind zinc and act as a molecular chaperone by stabilising the CtpC protein. Thus Rv3269 is renamed PacL1, for P-ATPase-associated chaperone-Like protein 1. Similarly, the CtpG and CtpV proteins are renamed PacL2 and PacL3. In the absence of PacL1, the CtpC protein is destabilised and degraded and M. tuberculosis becomes highly sensitive to zinc.
Ctp/PacL pairs are present in a large number of phylogenetically different bacteria. This discovery makes it possible to propose an entirely new model of resistance to metal stress in bacteria, through the intermediary of membrane platforms where both Ctp-type efflux pumps and small PacL molecular chaperones accumulate. These results open up numerous prospects not only for combating pathogenic bacteria by weakening their ability to resist metal stresses within their hosts, but also, more generally, for better understanding the metallobiology of prokaryotes.
Within the macrophages it parasitizes, the tuberculosis bacillus, M. tuberculosis, is stressed by the accumulation of metals (here zinc) in the phagocytosis vacuole. PacL family proteins (here PacL1) stabilise metal efflux pumps (here the CtpC pump) in membrane microdomains to resist metal intoxication. Fluorescence microscopy images show the co-localisation in the Mycobacterium plasma membrane of the PacL1 protein (blue) and the CtpC protein (yellow). ©Claire Lastrucci
Source
“Mycobacterial resistance to zinc poisoning requires assembly of P-ATPase-containing membrane metal efflux platforms”
Yves-Marie Boudehen, Marion Faucher, Xavier Maréchal, Roger Miras, Jérôme Rech, Yoann Rombouts, Olivier Sénèque, Maximilian Wallat, Pascal Demange, Jean-Yves Bouet, Olivier Saurel, Patrice Catty, Claude Gutierrez, Olivier Neyrolles
Nat Commun 2022 Aug 12 doi: 10.1038/s41467-022-32085-7
Researcher’s contact
Olivier Neyrolles | Olivier.Neyrolles@ipbs.fr | +33 (0)5 61 17 54 75
Press contact
IPBS : Francoise Viala | communication@ipbs.fr | +33 (0)6 01 26 52 59

