Mycobacterial Interactions with Host Cells

Group Leader
Our laboratory studies the complex interactions between Mycobacterium tuberculosis (Mtb) and the human host to identify new strategies for TB prevention and treatment. We investigate bacterial survival mechanisms, immune cell reprogramming—including macrophages, dendritic cells, T cells, and stromal cells—and TB/HIV-1 co-infection, using advanced imaging and multi-omics approaches to guide the development of host-directed therapies, vaccines, and antimicrobial strategies.
We study host-pathogen interactions in tuberculosis, with a special interest in deciphering immunity to TB and the links between M. tuberculosis evolution, physiology and virulence.

Our laboratory is dedicated to understanding the complex interplay between Mycobacterium tuberculosis (Mtb) and its human host, with the goal of developing new therapies and improving disease outcomes. We investigate how Mtb survives and thrives within macrophages and lung granulomas, uncovering the mechanisms that support its persistence under stress. Our research has revealed that toxin-antitoxin systems, metal efflux pumps, and nutrient acquisition pathways, including sulfate and cysteine metabolism, play critical roles in bacterial survival, redox balance, and virulence, representing promising therapeutic targets.
In parallel, we study how Mtb manipulates host immunity, shaping macrophage metabolism, dendritic cell function, and T cell responses to favor persistence. Our work has identified key pathways and compounds, such as the dendritic cell immunoreceptor (DCIR) and polysialic acid, that regulate inflammation and monocyte trafficking, while, stromal cells, innate lymphoid cells and CD4+ and CD8+ T cell diversity contribute to protective immunity.
Our investigations extend to TB and HIV-1 co-infection, revealing how Mtb promotes HIV-1 replication and dissemination through reprogrammed macrophages, highlighting potential interventions for co-infected patients.
Our laboratory combines advanced imaging, omics technologies, and collaborative expertise to study these processes at the cellular and organ level, aiming to translate our discoveries into host-directed therapies, improved vaccines, and antimicrobial strategies. By integrating insights into mycobacterial physiology and host immunity, our team seeks to illuminate TB biology and accelerate the development of innovative interventions to control this devastating disease.
Our research group brings together a vibrant and diverse community of scientists, including PhD students, postdoctoral fellows, early-career and senior researchers, as well as collaborators from France and around the world. We are committed to fostering an open, inclusive, and supportive environment that values diversity of backgrounds, perspectives, disciplines, and career paths.
Our fellows go on to thrive in leading laboratories and institutions, in both the public and private sectors, in France and internationally, reflecting the rigor of our training, the strength of our mentorship, and the breadth of opportunities we cultivate for their careers.
Together, we advance fundamental knowledge while training the next generation of scientists to tackle TB through multidisciplinary, collaborative, and globally engaged approaches.
Team members
Research Scientists
Pierre Dupuy (CNRS)
Claude Gutierrez (University)
Denis Hudrisier (University)
Fabien Letisse (University)
Geanncarlo Lugo (CNRS)
Olivier Neyrolles (CNRS)
Yannick Poquet (University)
Yoann Rombouts (CNRS)
Philippe Vogeleer (University)
Research Engineer
Annie Behar
Florence Levillain (CNRS)
Postdoctoral Fellows
Elizabeth Bautista
Saurabh Chugh
Tejan Lodhiya
PhD Students
Louis Benastre
Maxime Caouaille
Clara Deyts
Maxime Kaiser
Maxime Pingret
Stella Rousset
Research Projects
Co-PIs | Pierre DUPUY, Claude GUTIERREZ, Fabien LETISSE, Yannick POQUET, Philippe VOGELEER
Co-PIs | Denis HUDRISIER, Geanncarlo LUGO, Yoann ROMBOUTS
Selected Publications
Dupuy et al. (2026) Membrane-associated effluxosomes coordinate multi-metal resistance in Mycobacterium tuberculosis. The EMBO Journal
Le Mouëllic et al. (2025) Inorganic sulfate is critical for Mycobacterium tuberculosis lung tissue colonization and redox balance. Proc Natl Acad Sci USA
Boudehen*, Faucher* et al. (2022) Mycobacterial resistance to zinc poisoning requires assembly of P-ATPase-containing membrane metal efflux platforms. Nature Communications
Corral et al. (2022) ILC precursors differentiate into metabolically distinct ILC1-like cells during Mycobacterium tuberculosis infection. Cell Reports
Souriant et al. (2019) Tuberculosis exacerbates HIV-1 infection through IL-10/STAT3-dependent tunneling nanotube formation in macrophages. Cell Reports
Freire*, Gutierrez* et al. (2019) An NAD+ phosphorylase toxin triggers Mycobacterium tuberculosis cell death. Molecular Cell
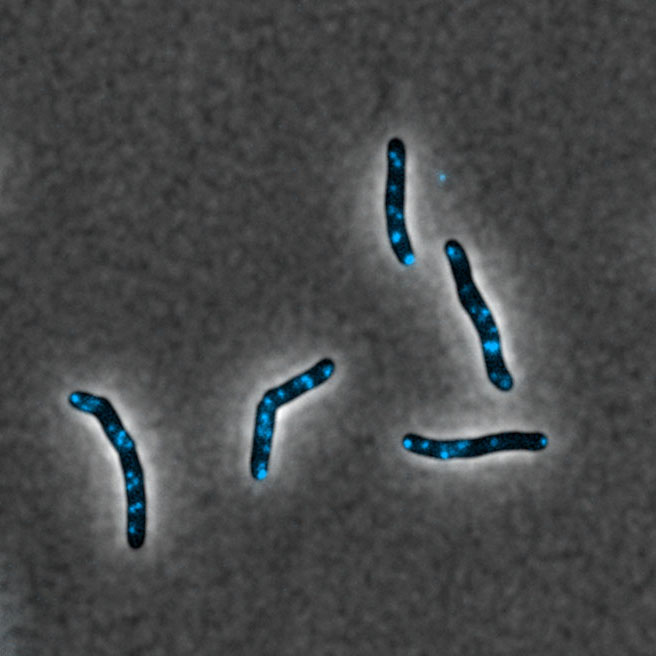
Confocal microscopy imaging of functional membrane micro domains (blue) in Mycobacterium tuberculosis © Y.-M. Boudehen
Our laboratory’s work has been widely recognized through national and international prizes, reflecting both scientific excellence and innovation in training the next generation of researchers. These distinctions honor the contributions of our team, from principal investigators to students and early-career scientists, highlighting our commitment to advancing TB research while fostering the development of future leaders in biomedical science.
April 2026 – Olivier Neyrolles is elected fellow of the European Academy of Microbiology
June 2024 – Wendy Le Mouëllic is national winner of the MT180 competition. Read more… (in French)
2022 – Olivier Neyrolles is elected member of the Academy of Sciences, Humanities, and Literature of Toulouse
October 2021 – Olivier Neyrolles is awarded the CNRS silver medal. Read more… (in French)
November 2020 – Olivier Neyrolles is awarded the Jacques Piraud Prize from the Fondation pour la Recherche Médicale.
2020 – Olivier Neyrolles is elected member of the European Academy (Academia Europaea), Biochemistry & Molecular biology section
October 2018 – The 2018 PEPS Prize for Pedagogical Innovation is awarded to the IMMUNOVA project (Active, interactive and flexible learning in immunology) led by Denis Hudrisier.
The IMMUNOVA project is composed of two axes: the first is the strong use of digital resources allowing the students to prepare themselves before and after the face-to-face sessions (also available online), which provides greater flexibility and learning autonomy for all students, especially those who cannot attend classes according to established schedules; for the second axis, the students are offered to conduct team projects in immunology according to different approaches (e.g., digital resource generation, interviews with researchers, 3D printing of proteins in FabLabs, conception of serious games).
Known more about the project – Watch the video! (in French)
December 2017 – Anthony Troegeler is awarded the Bretesche Prize in medicine from the Toulouse Academy of sciences.
Anthony did his PhD studies in our lab where he worked on the role of the innate immune receptor DCIR in immunity to tuberculosis. His work was published in January 2017 in Proc Natl Acad Sci USA. He will receive the Prize on December 3 at the Hôtel d’Assézat, Toulouse.
June 2017 – Yoann Rombouts is awarded the IGO Young Glycoscientist Award 2017.
December 2016 – Olivier Neyrolles receives the Sanofi-Institut Pasteur National Junior Award in the field of neglected tropical diseases for his research into tuberculosis, during a ceremony at the Institut Pasteur, Paris. Olivier and his team contributed to understanding several aspects of the complex interactions between the tuberculosis bacillus, Mycobacterium tuberculosis, and its host, including anti-mycobacterial immunity and mycobacterial evolution, physiology and virulence…
December 2014 – Alexandre Gouzy receives the Sanofi 2014 Prize from the Toulouse Academy of Sciences.
February 2014 – Olivier Neyrolles receives the “Coup d’élan pour la Recherche Française” Prize from the Bettencourt Schueller Foundation during a ceremony at the Institut de France, Paris, in which Madame F. Bettencourt Meyers presented the Prize. This Prize rewards teams specifically chosen for the promising nature of their research programs. The Prize recognises both the quality of the work of Olivier Neyrolles’ team and the quality of the research conducted in the Tuberculosis & Infection Biology Department at IPBS in the field of vaccine and antibiotics development.
April 2012 – Hélène Botella, winner of the AXA-Academy of Sciences prize based on her work in the involvement of zinc as a natural mechanism to fight against infections.
2009 – Olivier Neyrolles receives the CNRS Bronze medal.
Collaborations
A Unique Franco-Argentine Collaboration on TB Research
We are proud to highlight the unique and long-standing collaboration between our lab and the Instituto de Investigaciones Biomédicas en Retrovirus y Sida (INBIRS), led by Dr. Luciana Balboa, within the France–Argentina MAC-TB/HIV partnership. Launched in 2014 under the ECOS Sud program and formalized in 2016 as a Laboratoire International Associé (LIA) between IPBS and INBIRS, the LIA was coordinated by Olivier Neyrolles and Luciana Balboa. In 2021, the collaboration was renewed as a CNRS International Research Project (IRP), coordinated by Geanncarlo Lugo and Luciana Balboa. For over a decade, our teams have combined complementary expertise in TB immunometabolism and HIV biology to decipher how the tuberculous microenvironment reshapes macrophage lipid networks and metabolism, turning them into “Trojan horses” that facilitate HIV-1 persistence and dissemination. This highly productive partnership has generated major joint publications, trained numerous PhD students and postdoctoral fellows through sustained bilateral exchanges, and significantly increased the international visibility of our work on TB/HIV co-infection. The closing meeting held at IPBS on January 16, 2026, celebrated ten years of scientific achievements while reaffirming our shared commitment to deepen and expand this strategic Franco-Argentine collaboration in the years ahead.
We are indebtful to our many national and international collaborators, including:
- Samuel Behar University of Massachusetts Chan Medical School, Worcester, MA, USA
- Joel Ernst University of California, San Francisco, CA, USA
- Sho Yamasaki Research Institute for Microbial Diseases, Osaka, Japan
- David Sancho National Cardiovascular Research Centre, Madrid, Spain
- Patrice Catty Commissariat à l’Energie Atomique, Grenoble, France
- Ludovic Tailleux, Roland Brosch, Pierre Charneau & Lluis Quintana-Murci Institut Pasteur, Paris
- Matthias Willmans EMBL-Centre for Cellular and Molecular Imaging, Hamburg, Germany
- Manfred Wuhrer Leiden University Medical Centre, Leiden, The Netherlands
- Marcelo Kuroda University of California, Davis, CA, USA
- Luciana Balboa Academia Nacional de Medicina, Buenos Aires, Argentina
- Gerald Larrouy-Maumus Imperial College, London, UK
- Pierre Genevaux, CBI-LMGM, Toulouse, France
- Ting-Di Wu, Institut Curie, Paris, France
Funding
Our team is supported by several grants and fellowships from:
- European Union (FP7, H2020, Horizon Europe)
- ERA-NET JPI-AMR
- European Respiratory Society
- MSDAVENIR
- NIH
- Agence Nationale de la Recherche
- Agence Nationale de la Recherche sur le SIDA et les hépatites virales
- Fondation pour la Recherche Médicale
- Agence nationale de recherche sur le sida et les hépatites virales
- Fondation Bettencourt Schueller
- Fondation ARC pour la recherche sur le cancer
- Vaincre la Mucoviscidose
- Association Gregory Lemarchal
- Fondation Mérieux
- ECOS-Sud exchange programme (Argentina)
Our team was officially labelled by the Fondation pour la Recherche Médicale (2016-2019 and 2021-2024).
The complete list of our publications is available through Pubmed.
Preprints
TB vaccine research
Bettencourt, P.J.G., Joosten, S.A., Lindestam Arlehamn, C.S., Behr, M.A., Locht, C., Neyrolles, O. (2021) 100 years of the Bacillus Calmette-Guerin vaccine. Vaccine 39, 7221-7222 (View)
Levillain, F.*, Kim, H.*, Woong Kwon, K., Clark, S., Cia, F., Malaga, W., Lanni, F., Brodin, P., Gicquel, B., Guilhot, C., Bancroft, G.J., Williams, A., Jae Shin, S., Poquet, Y.**, Neyrolles, O.** (2020) Preclinical assessment of a new live attenuated Mycobacterium tuberculosis Beijing-based vaccine for tuberculosis. Vaccine 38, 1416-1423 (View)
Voss, G., Casimiro, D., Neyrolles, O., Williams, A., Kaufmann, S.H.E., McShane, H., Hatherill, M., Fletcher, H.A. (2018) Progress and challenges in TB vaccine development. F1000Res 7, 199 (View)
Kaufmann, S.H.E., Dockrell, H.M., Drager, N., Ho, M.M., McShane, H., Neyrolles, O., Ottenhoff, T.H.M., Patel, B., Roordink, D., Spertini, F., Stenger, S., Thole, J., Verreck, F.A.W., Williams, A., Consortium, T. (2017) TBVAC2020: Advancing Tuberculosis Vaccines from Discovery to Clinical Development. Front Immunol 8, 1203 (View)
Scriba, T.J., Kaufmann, S.H., Henri Lambert, P., Sanicas, M., Martin, C., Neyrolles, O. (2016) Vaccination Against Tuberculosis With Whole-Cell Mycobacterial Vaccines. J Infect Dis 214, 659-664 (View)
Immunity to TB, TB/HIV coinfection and other pathogens
Vahlas, Z.*, Deyts, C.*, Fried, S., Ben Neji, M., Pingret, M.**, Faivre, N.**, Monard, S.C.**, Hertel, Q., Maio, M., Barros, J., Lucas, A., Vu Manh, T.-P., Poincloux, R., Blanchet, F., Raynaud-Messina, B., Neyrolles, O., Lugo-Villarino G.#, Balboa, L.#, Vérollet, C.#. “Glycolysis inhibition during tuberculosis-driven metabolic rewiring reduces HIV-1 spread in macrophages.” Life Sci Alliance. 2026 In Press.
Lefrançais, E., Hudrisier, D., Neyrolles, O., Behar, S.M., Ernst, J.D. (2025) Finding and filling the knowledge gaps in mechanisms of T cell-mediated TB immunity to inform vaccine design. Nat Rev Immunol (View)
Bénard, A., Balboa, L., Caouaille, M., Ravon-Katossky, L., Meunier, E., Fillatreau, S., Sasiain, M.D.C., Neyrolles, O., Hudrisier, D. (2025) Human IL-6-Producing B Cells Promote the Differentiation of Monocytes Toward an Anti-Inflammatory CD16⁺CD163⁺CD206⁺PD-L1⁺ Phenotype in Tuberculosis. Eur J Immunol 55(4):e202451509
Maio, M., Joly, M., Vahlas, Z., Barros, J., Marín Franco, J.L., Genoula, M., Monard, S., Vecchione, M.B., Fuentes, F., Gonzalez Polo, V., Quiroga, M.F., Vermeulen, M., Argüello, R.J., Inwentarz, S., Musella, R., Ciallella, L., González Montaner, P., Palmero, D., Lugo Villarino, G., Sasiain, M.C., Neyrolles O., Verollet, C., Balboa, L. (2023) Elevated glycolytic metabolism of monocytes limits the generation of HIF-1α-driven migratory dendritic cells in tuberculosis. Elife (View)
Corral, D., Charton, A., Krauss, M.Z., Blanquart, E., Levillain, F., Lefrancais, E., Sneperger, T., Vahlas, Z., Girard, J.P., Eberl, G., Poquet, Y., Guery, J.C., Arguello, R.J., Belkaid, Y., Mayer-Barber, K.D., Hepworth, M.R., Neyrolles, O.**, Hudrisier, D.** (2022) ILC precursors differentiate into metabolically distinct ILC1-like cells during Mycobacterium tuberculosis infection. Cell Rep 39, 110715 (View)
Bernard-Raichon, L., Colom, A., Monard, S.C., Namouchi, A., Cescato, M., Garnier, H., Leon-Icaza, S.A., Metais, A., Dumas, A., Corral, D., Ghebrendrias, N., Guilloton, P., Verollet, C., Hudrisier, D., Remot, A., Langella, P., Thomas, M., Cougoule, C.**, Neyrolles, O.**, Lugo-Villarino, G**. (2021) A Pulmonary Lactobacillus murinus Strain Induces Th17 and RORgammat(+) Regulatory T Cells and Reduces Lung Inflammation in Tuberculosis. J Immunol 207, 1857-1870 (View)
Marin Franco, J.L., Genoula, M., Corral, D., Duette, G., Ferreyra, M., Maio, M., Dolotowicz, M.B., Aparicio-Trejo, O.E., Patino-Martinez, E., Charton, A., Metais, A., Fuentes, F., Soldan, V., Morana, E.J., Palmero, D., Ostrowski, M., Schierloh, P., Sanchez-Torres, C., Hernandez-Pando, R., Pedraza-Chaverri, J., Rombouts, Y., Hudrisier, D., Layre, E., Verollet, C., Maridonneau-Parini, I., Neyrolles, O., Sasiain, M.D.C., Lugo-Villarino, G., Balboa, L. (2020) Host-Derived Lipids from Tuberculous Pleurisy Impair Macrophage Microbicidal-Associated Metabolic Activity. Cell Rep 33, 108547 (View)
Dupont, M.*, Souriant, S.*, Balboa, L., Vu Manh, T.P., Pingris, K., Rousset, S., Cougoule, C., Rombouts, Y., Poincloux, R., Ben Neji, M., Allers, C., Kaushal, D., Kuroda, M.J., Benet, S., Martinez-Picado, J., Izquierdo-Useros, N., Sasiain, M.D.C., Maridonneau-Parini, I.**, Neyrolles, O.**, Verollet, C.**, Lugo-Villarino, G**. (2020) Tuberculosis-associated IFN-I induces Siglec-1 on tunneling nanotubes and favors HIV-1 spread in macrophages. Elife 9 (View)
Souriant, S., Balboa, L., Dupont, M., Pingris, K., Kviatcovsky, D., Cougoule, C., Lastrucci, C., Bah, A., Gasser, R., Poincloux, R., Raynaud-Messina, B., Al Saati, T., Inwentarz, S., Poggi, S., Morana, E.J., Gonzalez-Montaner, P., Corti, M., Lagane, B., Vergne, I., Allers, C., Kaushal, D., Kuroda, M.J., Sasiain, M.D.C., Neyrolles, O.**, Maridonneau-Parini, I.**, Lugo-Villarino, G.**, Verollet, C**. (2019) Tuberculosis Exacerbates HIV-1 Infection through IL-10/STAT3-Dependent Tunneling Nanotube Formation in Macrophages. Cell Rep 26, 3586-3599 e3587 (View) | This article was highlighted in Science
Dumas, A., Corral, D., Colom, A., Levillain, F., Peixoto, A., Hudrisier, D., Poquet, Y., Neyrolles, O. (2018) The Host Microbiota Contributes to Early Protection Against Lung Colonization by Mycobacterium tuberculosis. Front Immunol 9, 2656 (View)
Lugo-Villarino, G., Troegeler, A., Balboa, L., Lastrucci, C., Duval, C., Mercier, I., Benard, A., Capilla, F., Al Saati, T., Poincloux, R., Kondova, I., Verreck, F.A.W., Cougoule, C., Maridonneau-Parini, I., Sasiain, M.D.C., Neyrolles, O. (2018) The C-Type Lectin Receptor DC-SIGN Has an Anti-Inflammatory Role in Human M(IL-4) Macrophages in Response to Mycobacterium tuberculosis. Front Immunol 9, 1123 (View)
Benard, A., Sakwa, I., Schierloh, P., Colom, A., Mercier, I., Tailleux, L., Jouneau, L., Boudinot, P., Al-Saati, T., Lang, R., Rehwinkel, J., Loxton, A.G., Kaufmann, S.H.E., Anton-Leberre, V., O’Garra, A., Sasiain, M.D.C., Gicquel, B., Fillatreau, S.**, Neyrolles, O.**, Hudrisier, D.** (2018) B Cells Producing Type I IFN Modulate Macrophage Polarization in Tuberculosis. Am J Respir Crit Care Med 197, 801-813 (View)
Troegeler, A., Mercier, I., Cougoule, C., Pietretti, D., Colom, A., Duval, C., Vu Manh, T.P., Capilla, F., Poincloux, R., Pingris, K., Nigou, J., Rademann, J., Dalod, M., Verreck, F.A., Al Saati, T., Lugo-Villarino, G., Lepenies, B., Hudrisier, D., Neyrolles, O. (2017) C-type lectin receptor DCIR modulates immunity to tuberculosis by sustaining type I interferon signaling in dendritic cells. Proc Natl Acad Sci USA 114, E540-E549 (View)
Lastrucci, C., Benard, A., Balboa, L., Pingris, K., Souriant, S., Poincloux, R., Al Saati, T., Rasolofo, V., Gonzalez-Montaner, P., Inwentarz, S., Morana, E.J., Kondova, I., Verreck, F.A., Sasiain Mdel, C., Neyrolles, O.**, Maridonneau-Parini, I.**, Lugo-Villarino, G.**, Cougoule, C.** (2015) Tuberculosis is associated with expansion of a motile, permissive and immunomodulatory CD16(+) monocyte population via the IL-10/STAT3 axis. Cell Res 25, 1333-1351 (View)
Troegeler, A., Lastrucci, C., Duval, C., Tanne, A., Cougoule, C., Maridonneau-Parini, I.**, Neyrolles, O.**, Lugo-Villarino, G.** (2014) An efficient siRNA-mediated gene silencing in primary human monocytes, dendritic cells and macrophages. Immunol Cell Biol 92, 699-708
Lefevre, L.*, Lugo-Villarino, G.*, Meunier, E.*, Valentin, A., Olagnier, D., Authier, H., Duval, C., Dardenne, C., Bernad, J., Lemesre, J.L., Auwerx, J., Neyrolles, O., Pipy, B., Coste, A. (2013) The C-type lectin receptors dectin-1, MR, and SIGNR3 contribute both positively and negatively to the macrophage response to Leishmania infantum. Immunity 38, 1038-1049 (View)
Tanne, A., Ma, B., Boudou, F., Tailleux, L., Botella, H., Badell, E., Levillain, F., Taylor, M.E., Drickamer, K., Nigou, J., Dobos, K.M., Puzo, G., Vestweber, D., Wild, M.K., Marcinko, M., Sobieszczuk, P., Stewart, L., Lebus, D., Gicquel, B., Neyrolles, O. (2009) A murine DC-SIGN homologue contributes to early host defense against Mycobacterium tuberculosis. J Exp Med 206, 2205-2220 (View)
Tailleux, L., Pham-Thi, N., Bergeron-Lafaurie, A., Herrmann, J.L., Charles, P., Schwartz, O., Scheinmann, P., Lagrange, P.H., de Blic, J., Tazi, A., Gicquel, B., Neyrolles, O. (2005) DC-SIGN induction in alveolar macrophages defines privileged target host cells for mycobacteria in patients with tuberculosis. PLoS Med 2, e381 (View)
Tailleux, L., Schwartz, O., Herrmann, J.L., Pivert, E., Jackson, M., Amara, A., Legres, L., Dreher, D., Nicod, L.P., Gluckman, J.C., Lagrange, P.H., Gicquel, B., Neyrolles, O. (2003) DC-SIGN is the major Mycobacterium tuberculosis receptor on human dendritic cells. J Exp Med 197, 121-127 (View)
Maeda, N., Nigou, J., Herrmann, J.L., Jackson, M., Amara, A., Lagrange, P.H., Puzo, G., Gicquel, B., Neyrolles, O. (2003) The cell surface receptor DC-SIGN discriminates between Mycobacterium species through selective recognition of the mannose caps on lipoarabinomannan. J Biol Chem 278, 5513-5516 (View)
Mycobacterial evolution, physiology and virulence
Dupuy, P., Boudehen, Y.-M., Faucher, M., Buglino, J.A., Fay, A., Cantaloube, S., Grimoire, Y., Marcoux, J., Levet, F., Bettarel, L., Voisin, B., Rech, J., Bouet, J.-Y., Saurel, O., Sibarita, J.-B., Glickman, M., Gutierrez. C, Neyrolles, O. (2026) Membrane-associated effluxosomes coordinate multi-metal resistance in Mycobacterium tuberculosis. EMBO J (View)
Le Mouëllic, W., Levillain, F., Wu, T.D., Caouaille, M., Bousso, P., Poquet, Y., Neyrolles, O. (2025) Inorganic sulfate is critical for Mycobacterium tuberculosis lung tissue colonization and redox balance. Proc Natl Acad Sci USA 122, e2503966122
Chugh, S., Létisse, F., Neyrolles, O. (2025) The exometabolome as a hidden driver of bacterial virulence and pathogenesis. Trends Microbiol
Dupuy, P., Gutierez, C., Neyrolles, O. (2023) Modulation of bacterial membrane proteins activity by clustering into plasma membrane nanodomains. Mol Microbiol (View)
Boudehen, Y.M.*, Faucher, M.*, Marechal, X., Miras, R., Rech, J., Rombouts, Y., Seneque, O., Wallat, M., Demange, P., Bouet, J.Y., Saurel, O., Catty, P., Gutierrez, C., Neyrolles, O. (2022) Mycobacterial resistance to zinc poisoning requires assembly of P-ATPase-containing membrane metal efflux platforms. Nat Commun 13, 4731 (View)
Freire, D.M.*, Gutierrez, C.*, Garza-Garcia, A., Grabowska, A.D., Sala, A.J., Ariyachaokun, K., Panikova, T., Beckham, K.S.H., Colom, A., Pogenberg, V., Cianci, M., Tuukkanen, A., Boudehen, Y.M., Peixoto, A., Botella, L., Svergun, D.I., Schnappinger, D., Schneider, T.R., Genevaux, P., de Carvalho, L.P.S., Wilmanns, M., Parret, A.H.A.**, Neyrolles, O.** (2019) An NAD(+) Phosphorylase Toxin Triggers Mycobacterium tuberculosis Cell Death. Mol Cell 73, 1282-1291 e1288 (View)
Levillain, F.*, Poquet, Y.*, Mallet, L., Mazeres, S., Marceau, M., Brosch, R., Bange, F.C., Supply, P., Magalon, A., Neyrolles, O. (2017) Horizontal acquisition of a hypoxia-responsive molybdenum cofactor biosynthesis pathway contributed to Mycobacterium tuberculosis pathoadaptation. PLoS Pathog 13, e1006752 (View)
Neyrolles, O., Wolschendorf, F., Mitra, A., Niederweis, M. (2015) Mycobacteria, metals, and the macrophage. Immunol Rev 264, 249-263 (View)
Gouzy, A., Poquet, Y., Neyrolles, O. (2014) Nitrogen metabolism in Mycobacterium tuberculosis physiology and virulence. Nat Rev Microbiol 12, 729-737
Gouzy, A., Larrouy-Maumus, G., Bottai, D., Levillain, F., Dumas, A., Wallach, J.B., Caire-Brandli, I., de Chastellier, C., Wu, T.D., Poincloux, R., Brosch, R., Guerquin-Kern, J.L., Schnappinger, D., Sorio de Carvalho, L.P., Poquet, Y., Neyrolles, O. (2014) Mycobacterium tuberculosis exploits asparagine to assimilate nitrogen and resist acid stress during infection. PLoS Pathog 10, e1003928 (View)
Gouzy, A., Larrouy-Maumus, G., Wu, T.D., Peixoto, A., Levillain, F., Lugo-Villarino, G., Guerquin-Kern, J.L., de Carvalho, L.P., Poquet, Y., Neyrolles, O. (2013) Mycobacterium tuberculosis nitrogen assimilation and host colonization require aspartate. Nat Chem Biol 9, 674-676 (View)
Botella, H., Stadthagen, G., Lugo-Villarino, G., de Chastellier, C., Neyrolles, O. (2012) Metallobiology of host-pathogen interactions: an intoxicating new insight. Trends Microbiol 20, 106-112
Botella, H., Peyron, P., Levillain, F., Poincloux, R., Poquet, Y., Brandli, I., Wang, C., Tailleux, L., Tilleul, S., Charriere, G.M., Waddell, S.J., Foti, M., Lugo-Villarino, G., Gao, Q., Maridonneau-Parini, I., Butcher, P.D., Castagnoli, P.R., Gicquel, B., de Chastellier, C., Neyrolles, O. (2011) Mycobacterial p(1)-type ATPases mediate resistance to zinc poisoning in human macrophages. Cell Host Microbe 10, 248-259 (View)
Brodin, P., Poquet, Y.*, Levillain, F.*, Peguillet, I.*, Larrouy-Maumus, G., Gilleron, M., Ewann, F., Christophe, T., Fenistein, D., Jang, J., Jang, M.S., Park, S.J., Rauzier, J., Carralot, J.P., Shrimpton, R., Genovesio, A., Gonzalo-Asensio, J.A., Puzo, G., Martin, C., Brosch, R., Stewart, G.R., Gicquel, B., Neyrolles, O. (2010) High content phenotypic cell-based visual screen identifies Mycobacterium tuberculosis acyltrehalose-containing glycolipids involved in phagosome remodeling. PLoS Pathog 6, e1001100 (View)
Becq, J., Gutierrez, M.C., Rosas-Magallanes, V., Rauzier, J., Gicquel, B., Neyrolles, O.** Deschavanne, P.** (2007) Contribution of horizontally acquired genomic islands to the evolution of the tubercle bacilli. Mol Biol Evol 24, 1861-1871 (View)
Rosas-Magallanes, V., Deschavanne, P., Quintana-Murci, L., Brosch, R., Gicquel, B., Neyrolles, O. (2006) Horizontal transfer of a virulence operon to the ancestor of Mycobacterium tuberculosis. Mol Biol Evol 23, 1129-1135 (View)
A funny paper you may want to read…
Neyrolles, O., Hernandez-Pando, R., Pietri-Rouxel, F., Fornes, P., Tailleux, L., Barrios Payan, J.A., Pivert, E., Bordat, Y., Aguilar, D., Prevost, M.C., Petit, C., Gicquel, B. (2006) Is adipose tissue a place for Mycobacterium tuberculosis persistence? PLoS One 1, e43 (View)
We are extremely proud of our former research assistants, PhD students and post-doctoral fellows who are now continuing their career in the best labs, enterprises and institutions in life sciences!
- Wendy Le Mouëllic | Post-doctoral fellow, CIRI, Lyon, France
- Sarah Monard | Post-doctoral fellow, The Peter Doherty Institute, Melbourne, Australia
- Tamara Sneperger | Post-doctoral fellow, University of Edinburgh, UK
- Aurélien Boyancé | Project manager, Evotec, Toulouse, France
- Benjamin Raymond | Assistant professor, University of Newcastle, UK
- Yves-Marie Boudhen | Post-doctoral fellow, IRIM, Montpellier, France | Post-doctoral fellow, Institut Pasteur, Paris, France
- Alison Charton | Research assistant, Evotec, Toulouse, France
- Dan Corral | Post-doctoral fellow, NIAID, NIH, Bethesda, MD, USA
- Giulia Trimaglio | Post-doctoral fellow, Institute for Clinical Chemistry and Laboratory Medicine, Faculty of Medicine, Technische Universität Dresden, Germany
- Lucie Bernard-Raichon | Post-doctoral fellow, New York University, NY, USA | Post-doctoral fellow, IGFL, Lyon, France
- Maeva Dupont | Post-doctoral fellow, University of Oxford, UK
- Alexia Dumas | Post-doctoral fellow, University College Dublin, Ireland – Present: Post-doctoral fellow, IRSD, Toulouse, France
- Karine Pingris | Research engineer, Evotec, Toulouse, France
- André Colom | Research assistant, Evotec, Toulouse, France
- Danilo Pietretti | Project officer, European Commission
- Alan Bénard | Post-doctoral fellow, University Hospital, Erlangen, Germany
- Anthony Troegeler | Research associate, Evotec, Toulouse, France
- Anna Grabowska | Associate professor, Medical University of Warsaw, Warsaw, Poland
- Kanjana Nukdee | Professor, Ubon Ratchathani University, Thailand
- Ingrid Mercier | Business Project Leader, Airbus group, Toulouse, France
- Alexandre Gouzy | Post-doctoral fellow, now Assistant professor, Weill Cornell Medical College, NY, USA
- Hélène Botella | Post-doctoral fellow, Weill Cornell Medical College, NY, USA | Post-doctoral fellow, Imperial College, London, UK | Assistant professor, University of Toulouse, France
- Antoine Tanne | Executive Director of Translational Science | MPM BioImpact, Cambridge, Massachusetts, USA
- Jichan Jang | Professor, Gyeongsang National University, Jinju, South Korea
- Chongzhen Wang | Associate professor, Guilin Medical College, Guilin, China
- Chuan Wang | Lecturer, Fudan University, Shanghai, China

