Cystic fibrosis: organoid technology to identify new antimicrobial therapeutic strategies
Cystic fibrosis is the most common rare genetic disease. In these patients, respiratory infections contribute to the loss of lung function and are the leading cause of death. Among the pathogens associated with CF, nontuberculous mycobacteria such as Mycobacterium abscessus represent a major therapeutic challenge due to their natural resistance to antibiotics. Studying the interactions between the host and M. abscessus could help identify complementary antimicrobial therapeutic strategies. International research led by scientists at the Institute of Pharmacology and Structural Biology shows that activation of the NRF2-NQO1 antioxidant pathway in combination with a first-line antibiotic contributes to better control of M. abscessus infection. These results are published in PLOS Pathogens.
This research is based on organoid technology, which makes it possible to generate healthy or pathological mini-lungs in vitro. The organoids, obtained from patient biopsies, reproduce in vitro the architecture and function of the human airways, as well as the characteristics of this disease, for which the mouse model is irrelevant.
In this article, scientists show that organoids are a good model for reproducing the characteristics of M. abscessus infection, namely the formation of biofilms by the smooth S variant and cords by the rough R variant. Moreover, infection by M. abscessus induces the production of reactive oxygen species and epithelial cell death. At the molecular level, the infection by M. abscessus is associated with the activation of the NRF2-NQO1 axis, regulator of the antioxidant response, whose activation by the sulforaphane molecule reduces the bacterial charge in the organoids. Thus, M. abscessus benefits from this oxidizing environment for its development.
One of the advantages of organoid technology is its ability to mimic human pathologies such as cystic fibrosis in vitro, using patient biopsies. In this study, scientists show that patients’ organoids exhibit epithelial hyperplasia, exacerbated mucus secretion, and elevated oxidative stress associated with a defect in NRF2 expression, major features of cystic fibrosis. Infection of patient organoids with M. abscessus leads to exacerbated bacterial growth, increased oxidative stress and cell death, compared with healthy organoids. Thus, the cystic fibrosis environment favors the growth and virulence of M. abscessus. In this context, restoration of NRF2-NQO1 axis function by sulforaphane, alone or in combination with the first-line antibiotic cefoxitin, leads to improved infection control.
Pharmacological activation of the NRF2-NQO1 pathway could thus offer a therapeutic approach complementary to antibiotics for treating cystic fibrosis patients colonized by M. abscessus.
This research is funded by the Vaincre la Mucoviscidose foundation.
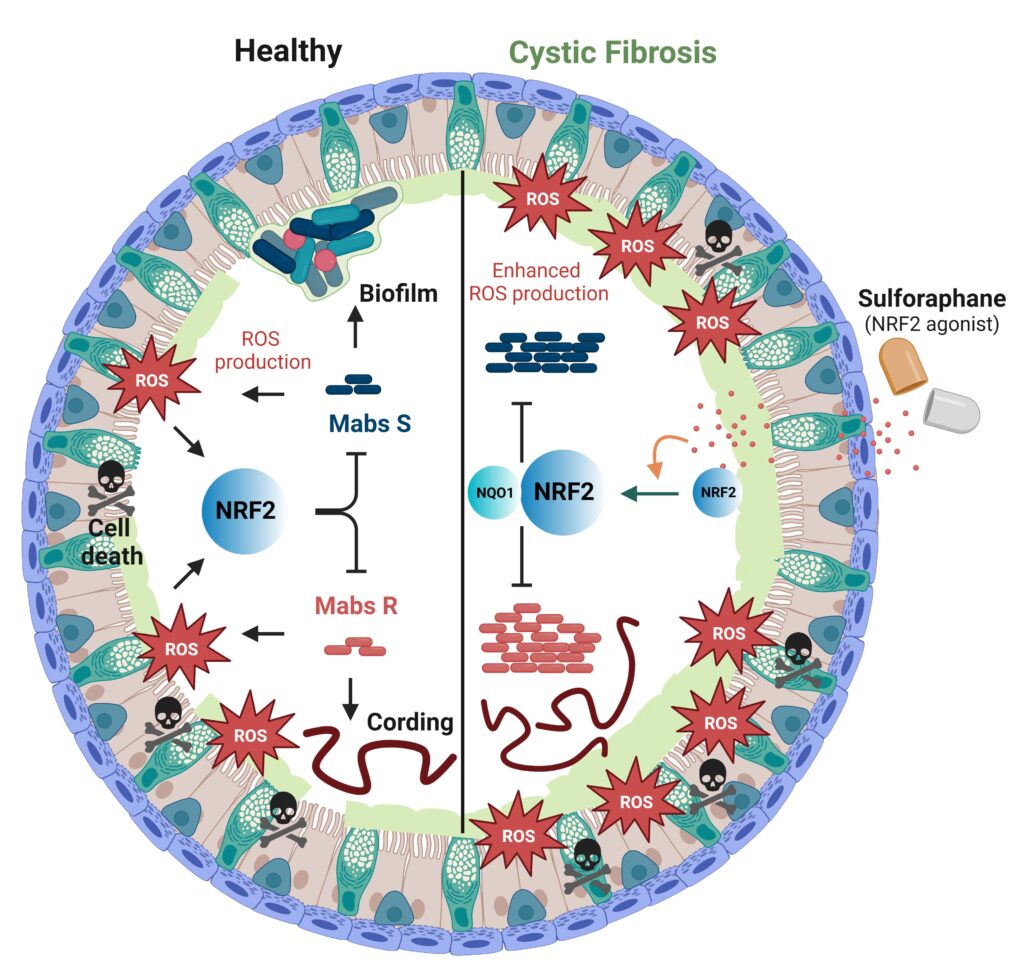
Mycobacterium abscessus exploits the oxidative environment of cystic fibrosis to multiply in its host.
In their basal state, healthy (H-AOs) and cystic fibrosis organoids (CF-AOs) exhibit similar cellular compositions. However, CF-AOs display key CF disease features: mucus buildup, increased oxidative stress, and elevated cell death. M. abscessus S and R forms replicate more efficiently in CF-AOs; S-M. abscessus generates biofilms, while R-M. abscessus forms cords, indicating higher virulence. M. abscessus infections triggers heightened oxidative stress in CF-AOs, exploiting the environment for growth. Enhancing detoxification pathways via NRF2 agonists, like sulforaphane, reduces ROS and cell death through NQO1 mRNA enrichment in CF-AOs, contributing to better M. abscessus growth control. Created in Biorender. © Céline Cougoule and Stephen Leon.
Source
Druggable redox pathways against Mycobacterium abscessus in cystic fibrosis patient-derived airway organoids.
SA Leon-Icaza, S Bagayoko, R Vergé, N Iakobachvili, C Ferrand, T Aydogan, C Bernard, A Sanchez Dafun, M Murris-Espin, J Mazières, P-J Bordignon, S Mazères, P Bernes-Lasserre, V Ramé, J-M Lagarde, J Marcoux, M-P Bousquet, C Chalut, C Guilhot, H Clevers, P J Peters, V Molle, G Lugo-Villarino, K Cam, L Berry, E Meunier, C Cougoule. PLOS Pathogens DOI: 10.1371/journal.ppat.1011559
Contact
Céline Cougoule (Celine.Cougoule@ipbs.fr)
Press
Françoise Viala (Communication@ipbs.fr)

