How PAXX keeps our DNA in check
Monitoring the integrity of DNA is the function of the many repair systems present in our cells, specialized in the detection and correction of specific anomalies. Among these, DNA double-strand breaks are particularly serious as they can lead to cell death. Hence, the lethal effect of double-strand breaks is exploited to eradicate cancer cells through radiotherapy and some chemotherapies. In a study published in Sciences Advances, scientists of IPBS and I2BC visualized the critical steps of the molecular assembly allowing cells to repair double-strand breaks in their DNA, in particular those involving the PAXX protein.
DNA is a fragile molecule that breaks easily, for example during air or space flights under the effect of cosmic radiation, during exposure to radioactivity or X-rays, or simply because cells grow in the presence of reactive oxygen. The dominant double-strand break repair system in human cells is the Non-Homologous End Joining (NHEJ), which coordinates many protein players to detect breaks, protect them, clean them up, and finally rejoin them. Among all the proteins involved, PAXX is the latest, identified in 2015 but whose role was not completely known. An international collaboration has just established how PAXX functions in NHEJ. When DNA is broken, the Ku protein very quickly encircles the ends of the break, thanks to its buoy-like shape, and serves as a mooring platform for subsequent proteins. Using two techniques that allow proteins to be visualized at the atomic scale (crystallography and cryo-electron microscopy), the researchers captured the moment when PAXX docks with Ku and precisely identified their contact areas. They showed that PAXX works in pairs with XLF, attached to Ku on the opposite side (Figure ). When these contact sites are destroyed by mutations, break repair is no longer possible and the cell dies. In addition to its fundamental aspect, this work opens therapeutic perspectives. Indeed, molecules that would block the assembly of PAXX and/or XLF on DNA breaks could increase the vulnerability of cancer cells to DNA double-strand breaks and ultimately increase the effectiveness of conventional treatments based on radiotherapy and certain chemotherapies.
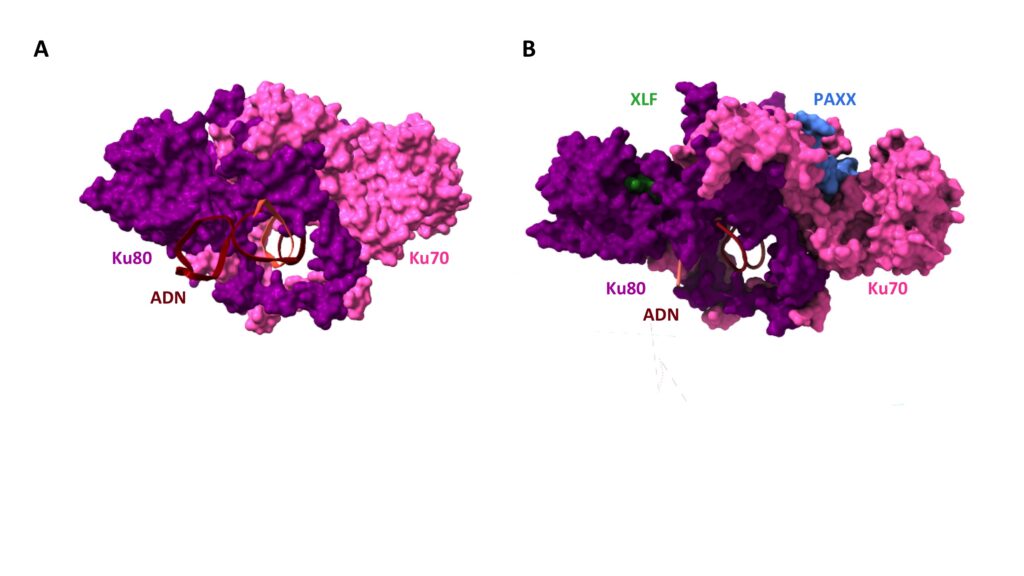
The Ku heterodimer, a flexible ring
A. Structure of Ku bound to DNA, determined by X-ray crystallography. DNA, in brown is bound to the Ku ring composed of the two subunits Ku80, in magenta and Ku70, in pink. B. Structure of Ku bound to two repair factors by their respective specific peptidic motif : XLF, in green on the Ku80 subunit. PAXX, in blue on the Ku70 subunit. Ku unfolds on both sides like a butterfly to accomodate binding of the two factors. © Seif-El-Dahan (I2BC)
Source
PAXX binding to the NHEJ machinery explains functional redundancy with XLF.
M Seif-El-Dahan1-†, A Kefala-Stavridi†, P Frit†, SW Hardwick†, DY Chirgadze, TM De Oliviera, S Britton, N Barboule, M Bossaert, AP Pandurangan, K Meek, TL Blundell, V Ropars*, P Calsou*, JB Charbonnier*, AK Chaplin*. Science Advances DOI: DOI: 10.1126/sciadv.adg2834. † co-first authors; * co-senior authors
Contact
Sébastien Britton (Sebastien.Britton@ipbs.fr), Patrick Calsou (Patrick.Calsou@ipbs.fr), Philippe Frit (Philippe.Frit@ipbs.fr)
Press contact
Françoise Viala (communication@ipbs.fr)

