Integrins in Immune Cells

Group Leader
Our team combines multi-omics approaches with functional perturbations in in vivo models to decode the dynamic regulation of integrin function in immune cells with potential for therapeutic intervention in cancer and inflammatory diseases. We focus our efforts in particular on the reprogramming of macrophages and the release of cytotoxic lymphocytes to restore an effective antitumor immune response.
We envision that a better understanding of the integrin-dependent interaction of immune cells with their environment will reveal new levers in the therapeutic arsenal against cancer.”

Immune cells are distinguished by their ability to gracefully patrol the body and cross boundaries to maintain tissue homeostasis and protect the host against pathogens. However, pathologies can arise when immune cells fail to eradicate pathogens and tumors or when they respond abnormally to healthy tissue. In all these cases, there is an urgent need to find new strategies to fine-tune the immune response to stimulate or suppress its activity. Thus, we study the complex and dynamic interactions of immune cells with their environment throughout their journey.
Integrins are a widely expressed family of cell adhesion receptors that participate in cellular interactions in many tissues. The role of integrins in immune cells is critical for their trafficking, activation, and function to shape successful immunity. Intensive research over the past four decades has unequivocally shown that integrins are attractive targets for recalibrating immune responses in local microenvironments. But still, direct targeting of their extracellular domain to interfere with their function has proven disappointing, in part because this method often fails to target a particular cell type, thus limiting the use of integrin antagonists. Instead, we intend to target intracellular signaling events that orchestrate integrin activities and which are far from being elucidated in immune cells. In this regard, we investigate the diversity and specificity of signaling pathways in various cellular contexts and investigate their potential to selectively manipulate specific subsets of immune cells for therapeutic purposes. This underappreciated strategy aligns with the emerging paradigm that signaling events in hematopoietic cells operate in both a cell-type and integrin-specific manner.
We mainly focus on integrin-dependent infiltration and immune cell function in the tumor microenvironment. Poor prognosis is largely correlated with the abundance of tumor-associated macrophages, as they promote disease progression and resistance to cancer therapies. Conversely, cytotoxic T lymphocytes struggle to penetrate in large numbers into the dense fibrous network of the tumor stroma, thus compromising their effectiveness in eliminating cancer cells. We strive to design novel strategies to restore antitumor immunity by manipulating the adhesive properties of immune cells to specifically reprogram their activity while selectively modulating their access to the solid tumor microenvironment.
Given the potential of adhesion molecules to uncouple multiple facets of immune cell functions, our research could have profound implications in many other pathological contexts beyond cancer, including inflammation and infectious disease.
Team members
Research Scientists
PhD Students
Marianna Plozza (50%)
Cédric Fabre (50%)
Research Engineer
Charlotte Payrault (IE INCa, 100%)
Lagarrigue et al. (2022) Direct binding of Rap1 to Talin1 and to MRL proteins promotes integrin activation in CD4+ T cells. J Immunol
Lee et al. (2022) Phostensin enables lymphocyte integrin activation and population of peripheral lymphoid organs. J Exp Med
Lagarrigue et al. (2020) Talin-1 is the principal platelet Rap1 effector of integrin activation. Blood
Gingras et al. (2019) Rap1 binding and a lipid-dependent helix in talin F1 domain promote integrin activation in tandem. J Cell Biol
Lagarrigue et al. (2017) Cutting Edge: Loss of T Cell RIAM Precludes Conjugate Formation with APC and Prevents Immune-Mediated Diabetes. J Immunol
Lagarrigue et al. (2015) A RIAM/lamellipodin-talin-integrin complex forms the tip of sticky fingers that guide cell migration. Nat Commun
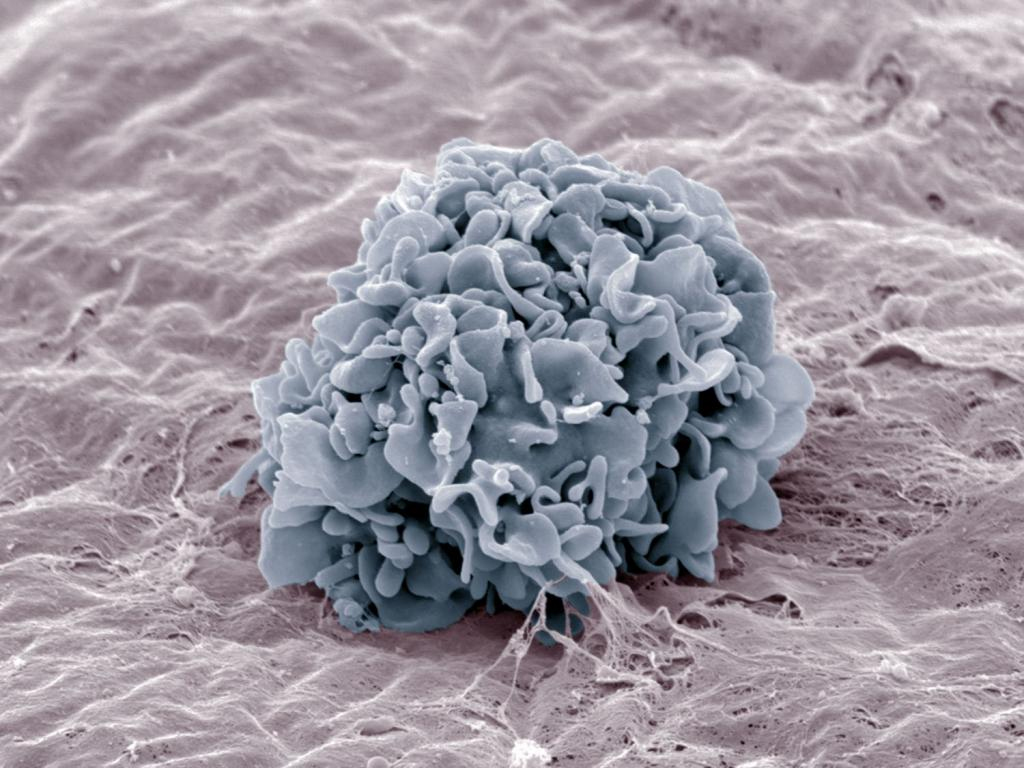
Scanning electron micrograph showing the adhesion of a macrophage onto the peritoneal basement membrane. © Renaud Poincloux
Here are our main recent publications. The complete list is available on PubMed.
2022
Lagarrigue, F., Tan, B., Du, Q., Fan, Z., Lopez-Ramirez, M.A., Gingras, A.R., Wang, H., Qi, W., Sun, H. (2022) Direct binding of Rap1 to Talin1 and to MRL proteins promotes integrin activation in CD4+ T cells. J Immunol 208, 1378-1388
Lee, H.S.*, Sun, H.*, Lagarrigue, F.*, Kim, S.H.J., Fox, J.W., Sherman, N.E., Gingras, A.R., Ginsberg, M.H. (2022) Phostensin enables lymphocyte integrin activation and population of peripheral lymphoid organs. J Exp Med 219: e20211637
Sun, H., Lagarrigue, F., Ginsberg, M.H. (2022) The connection between Rap1 and Talin1 in the activation of integrins in blood cells. Front Cell Dev Biol 10 :908622
2021
Lagarrigue, F., Gingras, A.R. (2021) Src-mediated phosphorylation of RIAM promotes integrin activation. Structure 296: 100675
Sun, H., Lagarrigue, F., Wang, H., Fan, Z., Lopez-Ramirez, M.A., Chang, J.T., Ginsberg, M.H. (2021) Distinct integrin activation pathways for effector and regulatory T cell trafficking and function. J Exp Med 218: e20201524
2020
Lagarrigue, F.*, Paul, D.S.*, Gingras, A.R.*, Valadez, A.J., Sun, H., Lin, J., Cuevas, M.N., Ablack, J.N., Lopez-Ramirez, M.A., Bergmeier, W., Ginsberg, M.H. (2020) Talin-1 is the principal platelet Rap1 effector of integrin activation. Blood 136, 1180-1190
Ablack, J.N., Ortiz, J., Bajaj, J., Trinh, K., Lagarrigue, F., Cantor, J.M., Reya, T., Ginsberg, M.H., MARCH proteins mediate responses to antitumor antibodies. J Immunol 205, 2883-2892
2019
Gingras, A.R.*, Lagarrigue, F.*, Cuevas, M.N., Valadez, A.J., Zorovich, M., McLaughlin, W., Lopez-Ramirez, M.A., Seban, N., Ley, K., Kiosses, W.B., Ginsberg M.H. (2019) Rap1 binding and a lipid-dependent helix in talin F1 domain promote integrin activation in tandem. J Cell Biol 218, 1799-1809
2018
Lagarrigue, F., Gingras, A.R., Paul, D.S., Valadez, A.R., Cuevas, M.N., Sun, H., Lopez-Ramirez, M.A., Goult, B.T., Shattil, S.J., Bergmeier, W., Ginsberg, M.H. (2018) Rap1 binding to the talin1 F0 domain makes a minimal contribution to murine platelet GPIIb-IIIa activation. Blood Adv 2, 2358-2368
Sun, H., Lagarrigue, F., Gingras, A.R., Fan, Z., Ley, K., Ginsberg, M.H. (2018) Transmission of integrin β7 transmembrane domain topology enables gut lymphoid tissue development. J Cell Biol 217, 1453-1465
2017
Lagarrigue, F., Gertler, F.B., Ginsberg, M.H., Cantor, J.M. (2017) Cutting Edge: Loss of T cell RIAM precludes conjugate formation with APC and prevents immune-mediated diabetes. J Immunol 198, 3410-3415
2016
Lagarrigue, F., Kim, C., Ginsberg, M.H. (2016) The Rap1-RIAM-talin axis of integrin activation and blood cell function. Blood 128, 479-487
2015
Lagarrigue, F.*, Vikas Anekal, P.*, Lee, H.S., Bachir, A.I., Ablack, J.N., Horwitz, A.F., Ginsberg, M.H. (2015) A RIAM/lamellipodin-talin-integrin complex forms the tip of sticky fingers that guide cell migration. Nat Commun 6:8492
2014
Ye, F.*, Lagarrigue, F.*, Ginsberg, M.H. (2014) SnapShot: talin and the modular nature of the integrin adhesome. Cell 156, 1340-1340.e1
We are indebted to our many collaborators including:
Claudine Blin – Laboratoire de PhysioMédecine Moléculaire, Nice, France
Jérôme Cros – Hôpital Beaujon, Paris, France
Mark Ginsberg – University of California San Diego, CA, USA
Christine Jean & Corinne Bousquet – Centre de Recherches en Cancérologie de Toulouse, France
Rémy Nicolle – Centre de Recherche sur l’Inflammation, Paris, France
Julie Pannequin – Institut de Génomique Fonctionnelle, Montpellier, France
Christel Verollet & Renaud Poincloux – Institut de Pharmacologie et de Biologie Structurale, Toulouse, France
Our team is and has been supported over the past five years by grants or scholarships from:
- ATIP-Avenir
- Institut National du Cancer
- Institut Thématiques Multi-Organismes (ITMO) – Cancer
- Marie Sklodowska-Curie Actions (H2020, Horizon Europe)
- Cancer, Aging & Rejuvenation Graduate School
- Fondation ARC pour la Recherche sur le Cancer
- Fondation de France
- Fondation Toulouse Cancer Santé
- La Ligue Nationale Contre le Cancer
November 2022 – Cédric Fabre was awarded by the Fonroga Foundation for his Master’s internship.
July 2022 – Thibaut Sanchez obtained the support of the ARC Foundation for cancer research for the financing of his fourth year of doctorate.
June 2022 – Marianna Plozza obtained a competitive 3-year doctoral scholarship from the Toulouse Graduate School of Cancer Aging and Rejuvenation (CARe).
April 2022 – Thibaut Sanchez received a travel grant from the French Society of Cellular Biology to attend the “8th meeting of the Invadosome Consortium: Pushing the frontiers of cellular adhesion and invasion”, held in Sète in October 2022.
October 2020 – Frédéric Lagarrigue has received the ATIP-Avenir label, which supports and enables talented young researchers to launch their own research team.
October 2019 – Thibaut Sanchez has received financial support from the National League Against Cancer for a 3-year doctoral scholarship.
July 2019 – Frédéric Lagarrigue received the first young researcher prize from the Fonroga Foundation.
November 2012 – Frédéric Lagarrigue received the Fernand Caujolles prize from the Academy of Sciences, Inscriptions and Beautiful Letters of Toulouse.

