Marine sponges inspire a new family of anticancer agents
A new generation of potential anticancer agents has been developed jointly by three labs in Toulouse. These molecules, which were bioinspired by compounds isolated from marine sponges, are now ongoing pre-clinical tests and open many prospects to treat cancer. This results are published in the Journal of Medicinal Chemistry.
To develop and defend themselves, marine organisms produce a wide variety of metabolites which play a critical role in their survival in the fiercely competitive marine environment. Over millions of years of evolution, these small organic molecules have acquired potent and selective properties, ranging from attraction to repulsion and even toxicity towards neighboring organisms. As such, researchers worldwide are actively exploring and attempting to exploit the chemical diversity of marine compounds.
Research efforts focused on compounds produced by marine sponges from the Petrosia species identified a set of polyacetylenic lipids with potent cytotoxic activity, which hold promise for cancer treatment. These lipids consist of a long lipid tail and of a terminal chiral# alkynylcarbinol motif (see the figure below). In order to evaluate and exploit their therapeutical value, a collaborative research program at the intersection of chemistry and biology was initiated a decade ago by teams from three research institutes in Toulouse##. Over this period, approximately 150 synthetic analogues of these natural polyacetylenic lipids were developed, with one of them demonstrating a cytotoxicity 1000 times greater than the reference natural product. This research program played a crucial role in identifying the structural elements that determine the cytotoxic activity of these lipids. Subsequently, the research teams discovered that the cytotoxicity of these compounds arises from their enantiospecific### bioactivation by a cellular enzyme which convert them into highly reactive species. Once bioactivated these lipids reacts with essential proteins to which they attach, thereby disrupting their biological function and localization and triggering a cascade of events ultimately resulting into the death of cancerous cells expressing this specific enzyme.
Building upon these results, scientists designed and synthesized a new generation of synthetic analogues by combining the terminal alkynylcarbinol motif with various (hetero)aromatic moieties integrated into the lipidic chain. Introducing an aromatic group offered novel possibilities for structural variations and consequently for modulating their resulting biological properties. This series proved more stable in biological media but also more selective towards the bioactivating enzyme than previous molecules. Finally, their stability and safety profile was evaluated in mice, paving the way for future investigations of their anticancer activity in mouse models. This work, published in the Journal of Medicinal Chemistry, underscores the power of long-term multidisciplinary collaborations between chemists and biologists in advancing the development of innovative small molecules for potential anticancer therapies.
# “Chiral” refers to a molecule having two distinct, non-superimposable mirror-image forms, known as enantiomers. These enantiomers can exhibit different biological properties.
## The Laboratoire de chimie de coordination (LCC, CNRS), the Institut de pharmacologie et biologie structurale (IPBS, CNRS/Université Toulouse Paul Sabatier) and the laboratoire de Synthèse et de physico-chimie de molécules d’intérêt biologique (SPCMIB, CNRS/Université Toulouse Paul Sabatier).
### “Enantiospecific” means “which concerns only one of the enantiomers of a chiral molecule”.
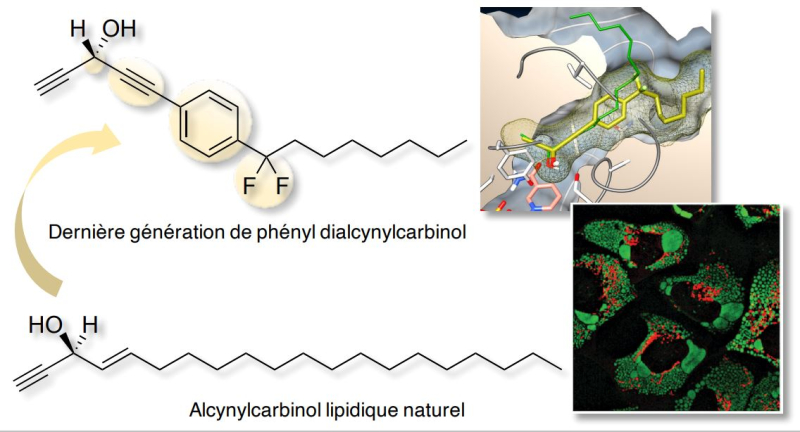
New generation of acetylenic lipid (top) and the reference natural molecule (bottom) comprising a chiral polar alkynylcarbinol motif in the terminal position. The new generation features an aromatic moiety which allows modulating the stability, selectivity and anticancer activity of this series of molecules © M. Bossuat et al.
Reference
Phenyl dialkynylcarbinols, a Bioinspired Series of Synthetic Antitumor Acetylenic Lipids
Margaux Bossuat, Pauline Rullière, Nadège Preuilh, Antonio Peixoto, Etienne Joly, Jean-Guillaume Gomez, Maroua Bourkhis, Frédéric Rodriguez, Fernanda Gonçalves, Isabelle Fabing, Hafida Gaspard, Vania Bernardes-Génisson, Valérie Maraval, Stéphanie Ballereau, Remi Chauvin*, Sébastien Britton* & Yves Génisson* J Med Chem 2023 Access the article
Contact (IPBS)
Sébastien Britton (Sebastien.Britton@ipbs.fr)
Press
Françoise Viala (Communication@ipbs.fr)

