Microenvironment, Cancer and Adipocytes

Group Leader
As obesity negatively impacts cancer survival, the main goal of our group is to characterize the role of tumor-surrounding adipocytes in cancer progression and the molecular mechanisms involved in both lean and obese conditions. Our team has been the first to define the specific phenotype of adipocytes within the tumor microenvironment, cells that we name Cancer-Associated Adipocytes (Dirat et al, Cancer Research, 2011, 800 citations).
Through their secretory and metabolic functions and their plasticity, adipocytes are key players in tumor progression.
Although AT share common features, each fat depot exhibits metabolic and secretory specificities. Thus, the use of human AT close to each type of tumor is key to the relevance of our results. The strong collaboration that we established since several years with clinicians allow us to work with human fat depots (mammary AT, periprostatic AT, bone-marrow AT). Some of the clinicians are members of the team and we welcome each year medical residents under training.
We have demonstrated that tumor-surrounding adipocytes promote aggressiveness by secreting soluble factors such as chemokines, pro-inflammatory cytokines and by modulating tumor cell metabolism. One of the most specific and emerging mechanism regarding the role of mature adipocytes in the tumor microenvironment involves the ability of cancer cells to advantageously exploit the nourishing role of adipocytes. At primary tumor sites, tumor cells induce lipolysis in adipocytes leading to the release free fatty acid (FFA) or contained in extra-cellular-vesicles that are taken up by tumor cells. These FFA trigger a complex metabolic remodeling in cancer cells increasing their survival, invasive and metastatic abilities as well as resistance to treatment. We are currently studying this metabolic crosstalk and its regulation by obesity in breast cancer using both human tumors samples (using groundbreaking approach of mass spectrometry imaging coupled to immunofluorescence) and in vitro 3D models (project 1). Another important aspect of our research is the study of this metabolic crosstalk at bone metastatic sites using bone marrow adipocytes in prostate cancer (project 2). Aside this metabolic crosstalk, in prostate cancer, we are investigating the role of abundant PPAT (that accumulates independently of body mass index) in prostate cancer progression, a project that highlights the central role of extra-cellular matrix remodeling (project 3).
The team possesses national and international recognition as assessed by our 5 highly cited papers (Top 1 % Web of Science in Clinical Science) in the field of adipose tissue and cancer (Dirat et al, Cancer Research, 2011 ; Bochet et al, Cancer Research, 2013 ; Lazar et al, Cancer Research, 2016 ; Laurent et al, Nature Communications, 2016 ; Wang et al, JCI Insight, 2017).
Team members
Research Scientists
Catherine Muller (University)
Camille Attané (CNRS)
Landry Blanc (CNRS)
Delphine Milhas (University)
Research Assistants
Stéphanie Dauvillier (University)
Mohamed Moutahir (CNRS)
Pascale Bernes-Lasserre
Clinicians
Charlotte Vaysse (Toulouse Hospitals)
Mathieu Roumiguié (Toulouse Hospitals)
PhD Students
Caroline Bouche
Marine Hernandez
Mathilde Lacombe
Camille Monchatre
Alessandro Taccini
Master Students
Adrien Leeman
Margaux Renier
Roumiguié, Estève et al. (2022) Periprostatic adipose tissue displays a chronic hypoxic state that limits its expandability Am J Pathol
Attané et al. (2020) Human bone marrow is comprised of adipocyte with specific lipid metabolism. Cell Rep
Clement E et al. (2020) Adipocyte extracellular vesicles carry enzymes and fatty acids that stimulate mitochondrial metabolism and remodeling in tumor cells. EMBO J
Lehuédé et al. (2019) Adipocytes promote breast cancer resistance to chemotherapy, a process amplified by obesity: role of the major vault protein (MVP). Breast Cancer Res
Wang YY et al. (2017) Mammary adipocytes stimulate breast cancer invasion through metabolic remodeling of tumor cells. JCI Insight (Top 1% Web of science)
Laurent et al. (2016) Periprostatic adipose tissue acts as a driving force for the progression of prostate cancer in obesity. Nat Commun (Top 1% Web of Science)
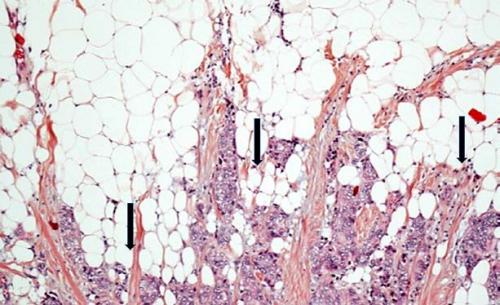
Section of invasive breast tumor (mauve) coming into contact of mature adipocytes (white discs). The adipocytes at close proximity of cancer cells exhibit a decrease in size and lipid content (arrows).
Collaborations
Research Laboratories
- Dr Anne BOULOUMIE, Institute of Cardiovascular and Metabolic Diseases, TOULOUSE, France
- Pr Olivier PIOT, Translational Biospectroscopy, REIMS, France
- Dr Marc POIROT, Cancer Research Institute, TOULOUSE, France
- Pr Philippe VALET and Dr Cedric DRAY, Institute Restore, TOULOUSE, France
- Pr Philip SCHERER, Touchstone Diabetes Center, DALLAS, USA
- Dr Daniela QUAIL, Mac Gill university, MONTREAL, Canada
Translational Programs
- For our prostate cancer topic, a strong collaboration has been established with the Urology Department (Pr Mathieu ROUMIGUIÉ and colleagues) and the Pathology Department (Dr Sarah Pericart) of the Toulouse Cancer Institute.
- For our breast cancer topic, we are collaborating with the Breast Cancer Surgery Department (Pr Charlotte VAYSSE and colleagues) as well as the Pathology Department (Dr Camille FRANCHET) of the Toulouse Cancer Institute.
- For our program on bone metastasis, a collaboration has been established with the Orthopedic surgery Department (Pr Nicolas REINA).
Funding
Our team is/was supported by several grants and fellowships from:
- Association pour la Recherche sur les Tumeurs Prostatiques
- Association Française de Recherche sur l’Obésité
- Cancéropôle Grand Sud-Ouest
- Fondation ARC pour la Recherche sur le Cancer
- Fondation de France
- Fondation pour la Recherche Médicale
- Fondation Toulouse Cancer-Santé
- French National Institute against Cancer (INCA PL-BIO2016 and 2020)
Our team is officially labelled by the Ligue Nationale contre le Cancer (2020-2025)
The complete list of our publications is available through Pubmed.

